Amino Acid Chart With Pka
Amino Acid Chart With Pka - Consequently, they are fundamental components of our bodies and vital for physiological functions such as protein synthesis, tissue repair and nutrient absorption. Web the isoelectric point of an amino acid is the ph at which the amino acid has a neutral charge. At neutral ph the amino group is protonated, and the carboxyl group is deprotonated. Carbon, hydrogen, oxygen, nitrogen and in some cases sulphur. Web table \(\pageindex{2}\) shows the standard pk a values for the amino acids and can be used to predict the ionization/charge status of amino acids and their resulting peptides/proteins. Web most biochemistry courses will require you to know the following: See if you can calculate the isoelectric points. Summary of pkas of amino acids Amino acids are the building blocks that form polypeptides and ultimately proteins. Web amino acids are the building blocks of proteins. Web table of pka and pi values. Web most biochemistry courses will require you to know the following: At ph values above or below the isoelectric point, the molecule will have a net charge which depends on its pi value as well as the ph of the solution in which the amino acid is found. We will also discuss zwitterions,. Web amino acids reference chart. Amino acids are the building blocks that form polypeptides and ultimately proteins. Properties of common amino acids. Created by tracy kim kovach. Figure 2.13 shows the metabolic fates of catabolism of each of the amino acids. Amino acids are the building blocks that form polypeptides and ultimately proteins. Conjugate acid of − nh 2, i.e., − nh + 3 has pk a ~10. The isoelectric points range from 5.5 to 6.2. Web the isoelectric point of an amino acid is the ph at which the amino acid has a neutral charge. Web the pka is a. Figure 2.13 shows the metabolic fates of catabolism of each of the amino acids. Properties of common amino acids. Conjugate acid of − nh 2, i.e., − nh + 3 has pk a ~10. The isoelectric points range from 5.5 to 6.2. Web here is a table with some structures and pk a values for amino acids. Web the pka is a measure of the strength of an acid, i.e., the lower the pk a stronger the acid. Properties of common amino acids. The isoelectric points range from 5.5 to 6.2. Web table \(\pageindex{2}\) shows the standard pk a values for the amino acids and can be used to predict the ionization/charge status of amino acids and. Web most biochemistry courses will require you to know the following: Carbon, hydrogen, oxygen, nitrogen and in some cases sulphur. Web table of pka and pi values. The r group, which differs for each amino. You will learn how to calculate the isoelectric point, and the effects of ph on the amino acid's overall charge. Properties of common amino acids. At ph values above or below the isoelectric point, the molecule will have a net charge which depends on its pi value as well as the ph of the solution in which the amino acid is found. We will also discuss zwitterions, or the forms of amino acids that dominate at the isoelectric point. See. You will learn how to calculate the isoelectric point, and the effects of ph on the amino acid's overall charge. Web amino acids are the building blocks of proteins. Web here is a table with some structures and pk a values for amino acids. They contain an amino group, carboxylic acid group, alpha carbon, and side chain. Titration curves show. Web table of pka and pi values. Web each amino acid has its own pi value based on the properties of the amino acid. The r group, which differs for each amino. Web the isoelectric point of an amino acid is the ph at which the amino acid has a neutral charge. At ph values above or below the isoelectric. We will also discuss zwitterions, or the forms of amino acids that dominate at the isoelectric point. Web amino acids are the building blocks of proteins. Most amino acids have a chiral carbon, which allows them to rotate polarized light. 3 pkx is the negative of the logarithm of the dissociation constant for any other group in the molecule. Web. Web amino acid pka c pka n pka r pi; Titration curves show the neutralization of these acids by added base, and the change in ph during the titration. Discover our full product line of amino acids, including alanine, isoleucine, leucine, valine, phenylalanine, tryptophan, tyrosine, aspargine, cysteine, glutamine, methionine, serine, threonine, aspartic acid. At ph values above or below the isoelectric point, the molecule will have a net charge which depends on its pi value as well as the ph of the solution in which the amino acid is found. Web here is a table with some structures and pk a values for amino acids. You will learn how to calculate the isoelectric point, and the effects of ph on the amino acid's overall charge. Figure 2.13 shows the metabolic fates of catabolism of each of the amino acids. Web amino acid reference chart contains the twenty amino acids found in eukaryotes, grouped according to their side chains and charge. The isoelectric points range from 5.5 to 6.2. We will also discuss zwitterions, or the forms of amino acids that dominate at the isoelectric point. Web most biochemistry courses will require you to know the following: Conjugate acid of − nh 2, i.e., − nh + 3 has pk a ~10. At neutral ph the amino group is protonated, and the carboxyl group is deprotonated. Web all amino acids have the same basic structure, which is shown in figure 2.1. They contain an amino group, carboxylic acid group, alpha carbon, and side chain. Web the amino acids themselves are constructed from a combination of the following elements: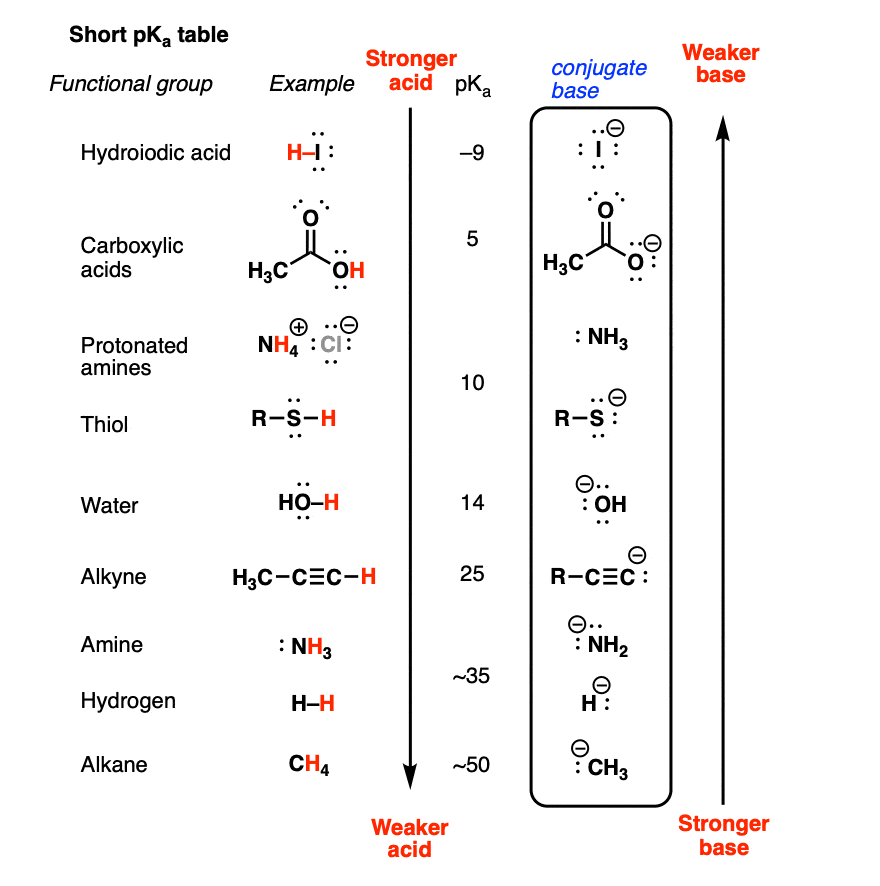
How To Use a pKa Table
AcidBase Equilibrium Part 1 How to Use the pKa Table — Organic

Pka Chart Of Amino Acids
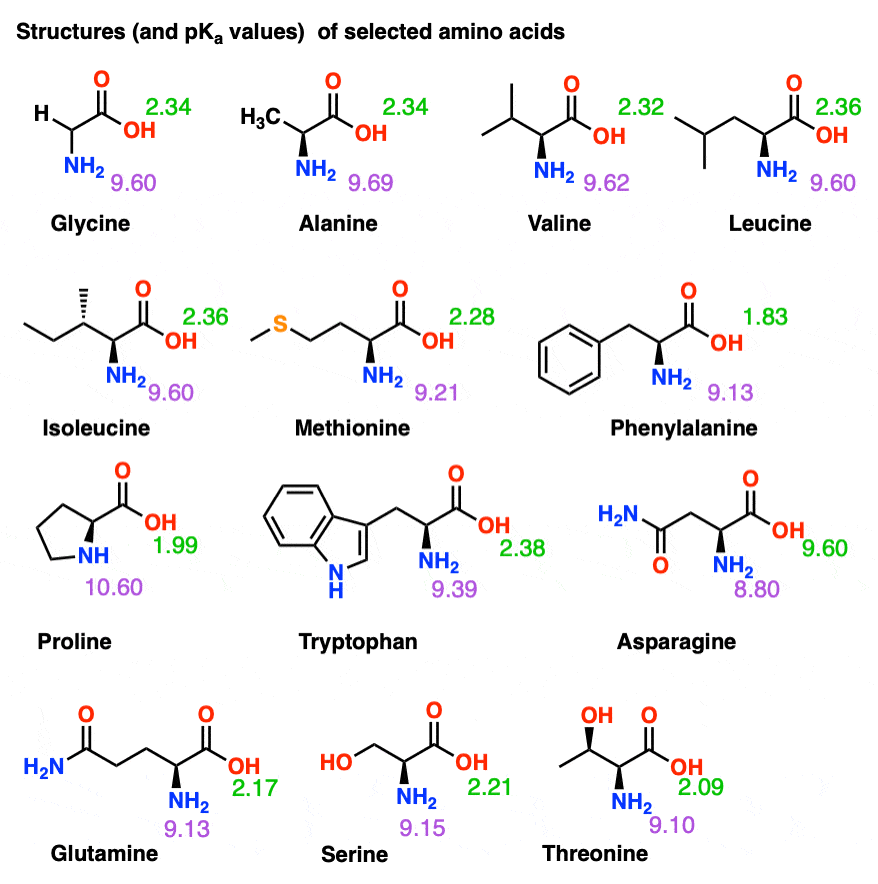
Isoelectric Points of Amino Acids (and How To Calculate Them) Master
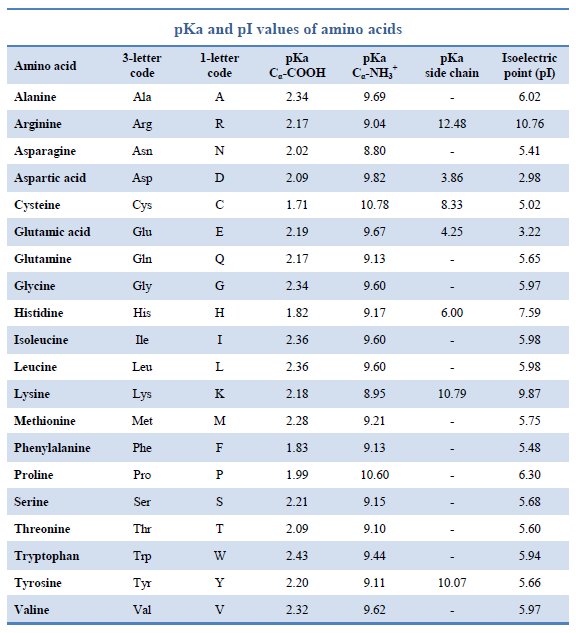
Amino acid properties
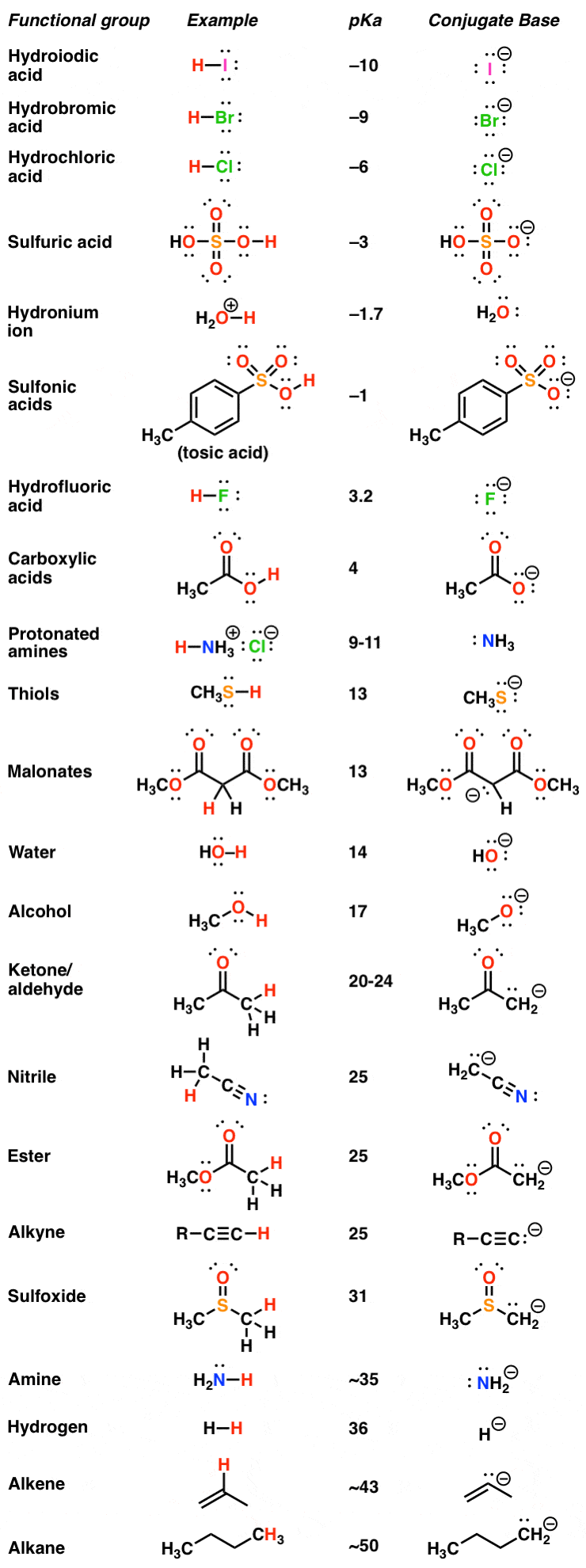
The pKa Table Is Your Friend
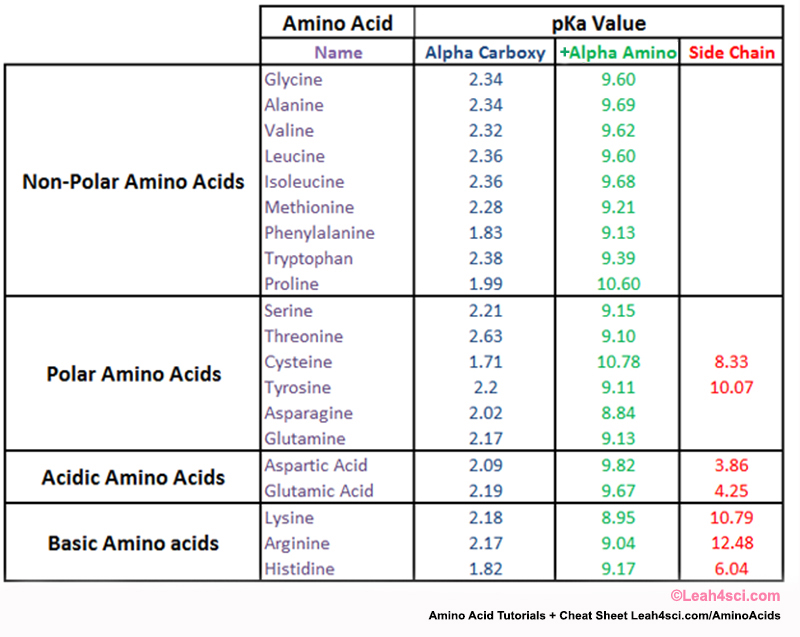
Amino Acid Charge in Zwitterions and Isoelectric Point MCAT Tutorial
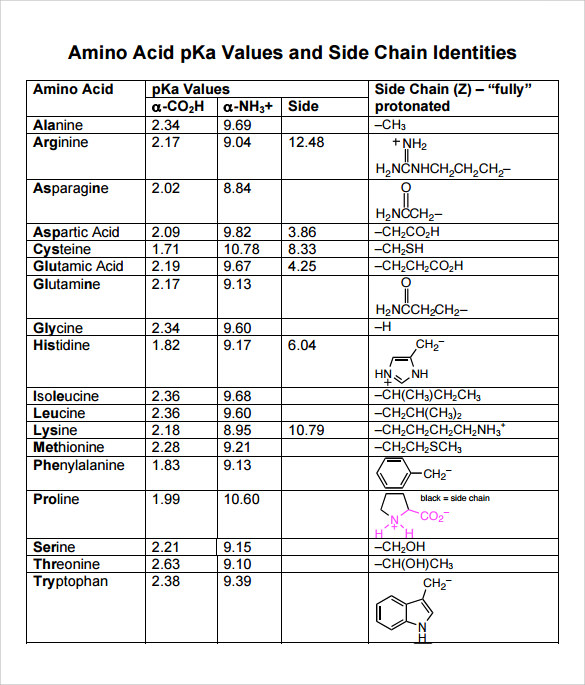
Pka Chart Amino Acids
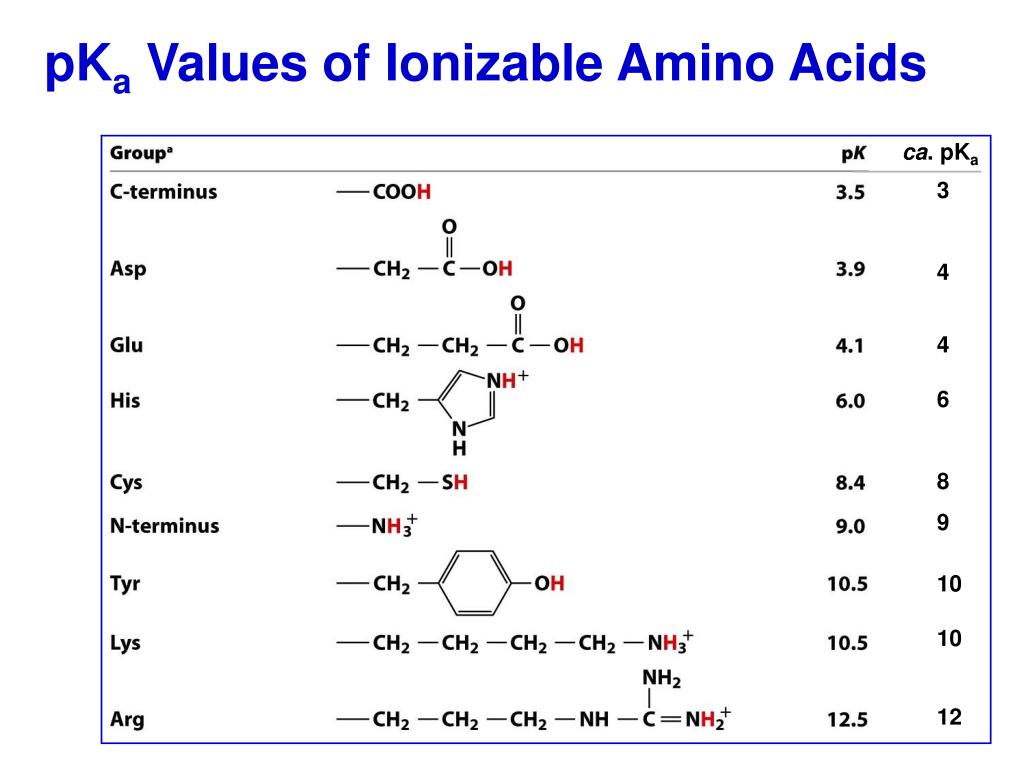
Amino Acids Pka Chart
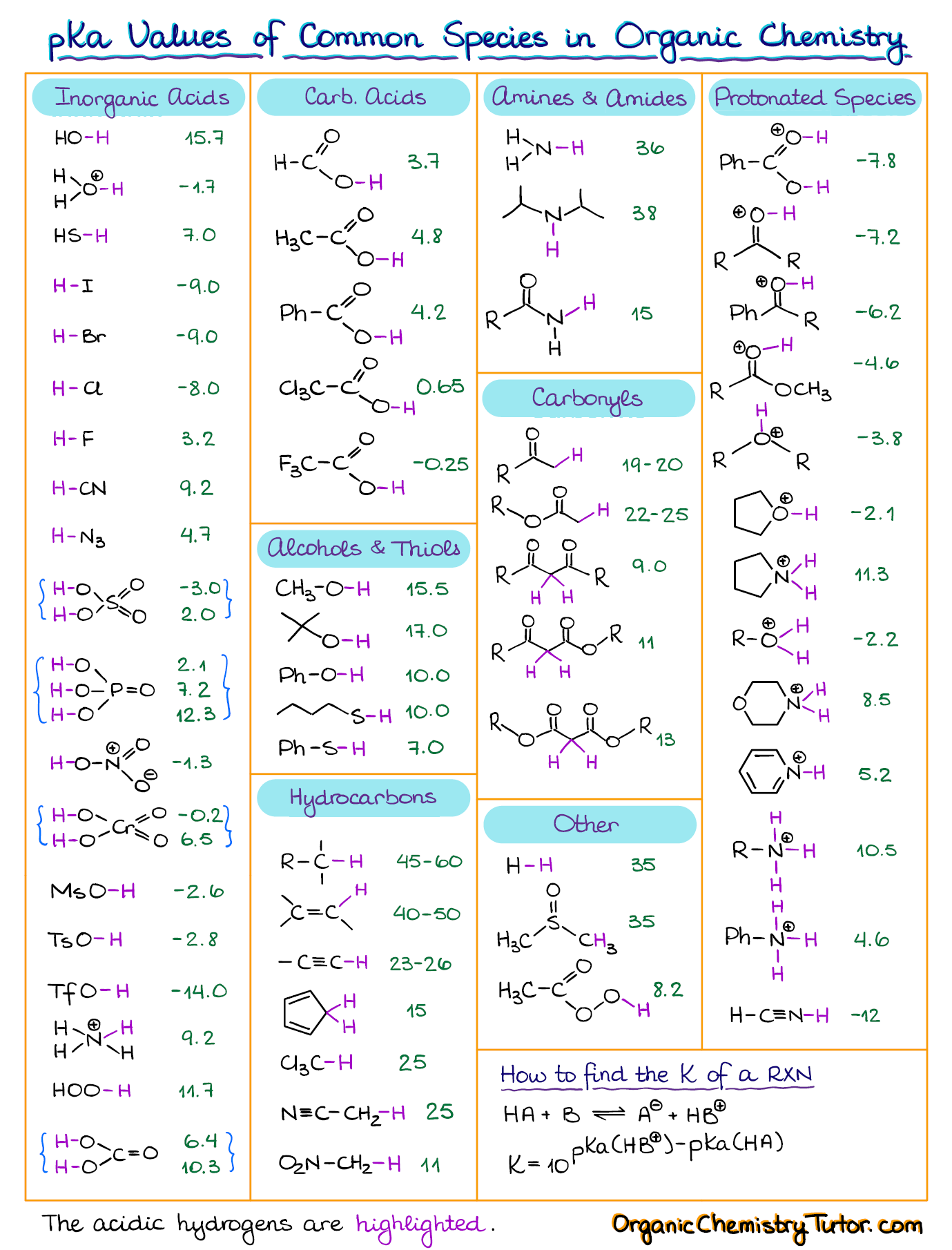
![[Infographic] Comprehensive pKa Chart r/chemistry](https://external-preview.redd.it/K3Snfd3HbLKkQbUsJ8g5GMVBN8te4Altg0_bder8QLE.jpg?auto=webp&s=d6f9b865541ac9cf9c578f5146e380a014396caa)
[Infographic] Comprehensive pKa Chart r/chemistry
Web Amino Acids Are The Building Blocks Of Proteins.
Amino Acids Are The Building Blocks That Form Polypeptides And Ultimately Proteins.
Created By Tracy Kim Kovach.
Carbon, Hydrogen, Oxygen, Nitrogen And In Some Cases Sulphur.
Related Post: