Atomic Radius Periodic Table Chart
Atomic Radius Periodic Table Chart - Web when you move right across a period, you’re adding more protons and electrons, but you stay at the same number of shells, rather than forcing electrons into higher shells, allowing the magnetic attraction between the protons and electrons to keep the radius of the atom smaller. Web june 18, 2023 by jay rana. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Below mentioned radii are the van der waals radius in picometer (pm)). Web major periodic trends include: This is due to the way electrons form shells around the nucleus. Web explore how atomic radius changes with atomic number in the periodic table of elements via interactive plots. The following table shows atomic radii computed from theoretical models, as published by enrico clementi and others in 1967. What is atomic radius on a periodic table? Web the periodic table of the elements (including atomic radius) element name. Web atomic radius is the distance from the atom’s nucleus to the outermost electron orbital, and a lot of trends in the periodic table rely on this property due to its relationship to other atomic properties such as nuclear charge and shielding. Web select from the following links to see visual periodicity representations for atomic radii, covalent radii, and van. Web when you move right across a period, you’re adding more protons and electrons, but you stay at the same number of shells, rather than forcing electrons into higher shells, allowing the magnetic attraction between the protons and electrons to keep the radius of the atom smaller. Pdf without crop marks | pdf with crop marks. Web atomic radii vary. Ionic radii are also available. Francium has the largest atomic size on the periodic table, and helium has the smallest atomic size. This is a periodic table with atomic radius values mentioned on it. These related values display the same trend in the periodic table. Web atomic radius trends on periodic table (video) | khan academy. Web atomic radius (ionic radius) atomic radius is the distance from the nucleus to the outermost stable electron while ionic radius is half the distance between two atomic nuclei that are just touching each other. In general, atomic radius or atom size decreases as you move from left to right. Web the atomic radius is the size of the atom,. In general, atomic radius or atom size decreases as you move from left to right. Web this table shows how the atom size, and atomic radius values change as you move horizontally and vertically across the periodic table. Web the periodic table contains nist’s latest critically evaluated data for atomic properties of the elements. This is due to the way. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Francium has the largest atomic size on the periodic table, and helium has the smallest atomic size. Web atomic radii vary in a predictable and explicable manner across the periodic table. The atomic radius of. Web when you move right across a period, you’re adding more protons and electrons, but you stay at the same number of shells, rather than forcing electrons into higher shells, allowing the magnetic attraction between the protons and electrons to keep the radius of the atom smaller. Web the trend on a graph. What is atomic radius on a periodic. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! This is due to the way electrons form shells around the nucleus. This is a periodic table with atomic radius values mentioned on it. Web explore how atomic radius changes with atomic number in the. The above atomic radii of the elements are the van der waals radius and they are in picometers (pm). Francium has the largest atomic size on the periodic table, and helium has the smallest atomic size. Web the atomic radius is the size of the atom, typically measured by the distance from the nucleus of the atom to the electron. Web atomic radius (ionic radius) atomic radius is the distance from the nucleus to the outermost stable electron while ionic radius is half the distance between two atomic nuclei that are just touching each other. Web in the periodic table, atomic radii decrease from left to right across a row and increase from top to bottom down a column. Web. Web june 18, 2023 by jay rana. Web atomic radius of all the elements are mentioned in the chart below. Below mentioned radii are the van der waals radius in picometer (pm)). In general, atomic radius or atom size decreases as you move from left to right. The atomic radius of atoms generally decreases from left to right across a period. Web explore how atomic radius changes with atomic number in the periodic table of elements via interactive plots. Atomic radii can be obtained from quantum mechanical calculations or. For instance, the radii generally decrease rightward along each period (row) of the table, from the alkali metals to the noble gases; Web select from the following links to see visual periodicity representations for atomic radii, covalent radii, and van der waals radii. Web atomic radius is the distance from the atom’s nucleus to the outermost electron orbital, and a lot of trends in the periodic table rely on this property due to its relationship to other atomic properties such as nuclear charge and shielding. Web in the periodic table, atomic radii decrease from left to right across a row and increase from top to bottom down a column. Web the atomic radius is the average distance from the center of the nucleus of a neutral atom to the outer boundary of its electron shell. Covalent radius can be calculated by measuring the distance between the two nucleus of two atoms in covalent compound. Ionic radii are also available. The following table shows atomic radii computed from theoretical models, as published by enrico clementi and others in 1967. These related values display the same trend in the periodic table.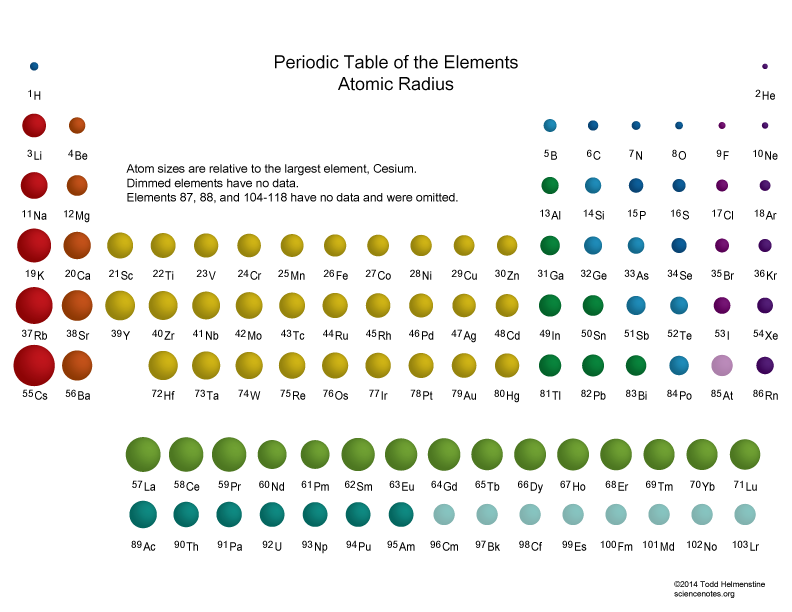
Atomic Radius Trends of the Periodic Table
/accurate-illustration-of-the-periodic-table-82020791-57cc76f23df78c71b66efbd7.jpg)
Atomic Radius Table Pdf Elcho Table
PPT Periodic Table PowerPoint Presentation, free download ID2617702

Atomic Radius of Elements
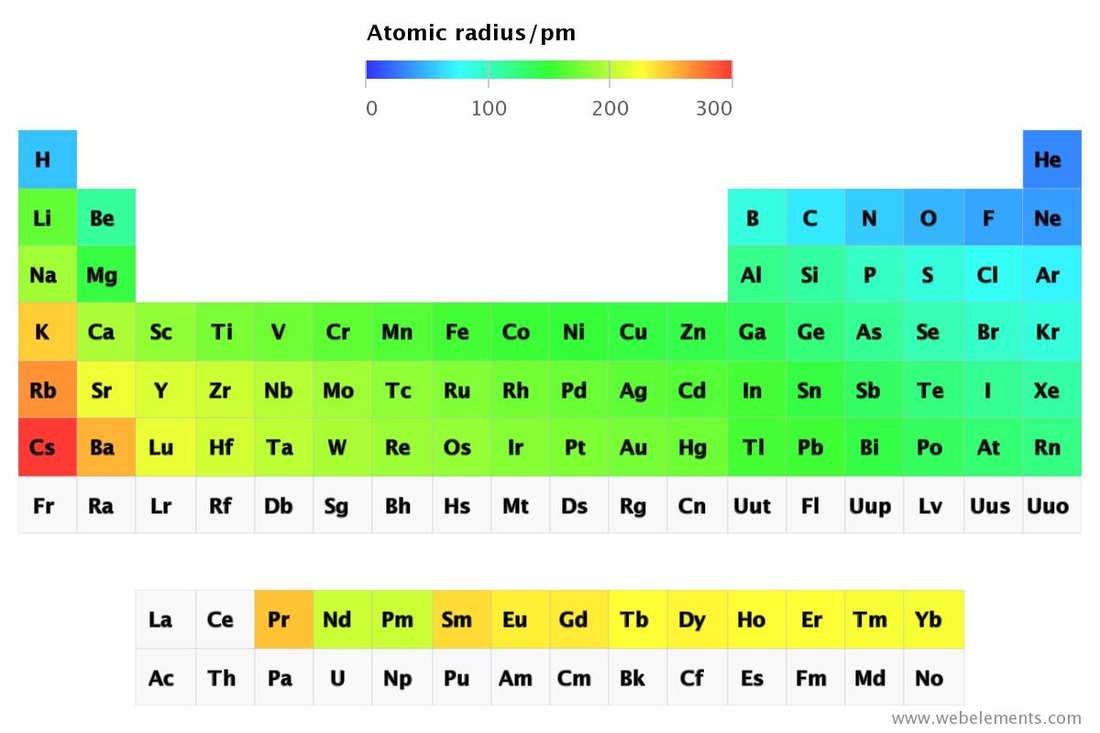
Atomic Radius of Elements The Periodic Table
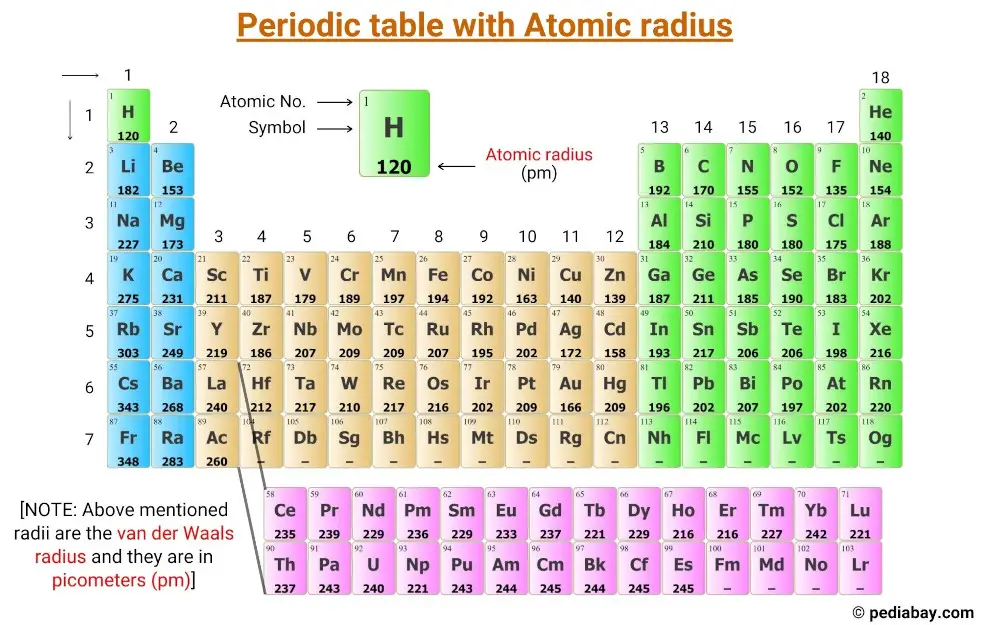
Atomic Radius of Elements (With Periodic table Chart) Pediabay
.png)
CK12Foundation
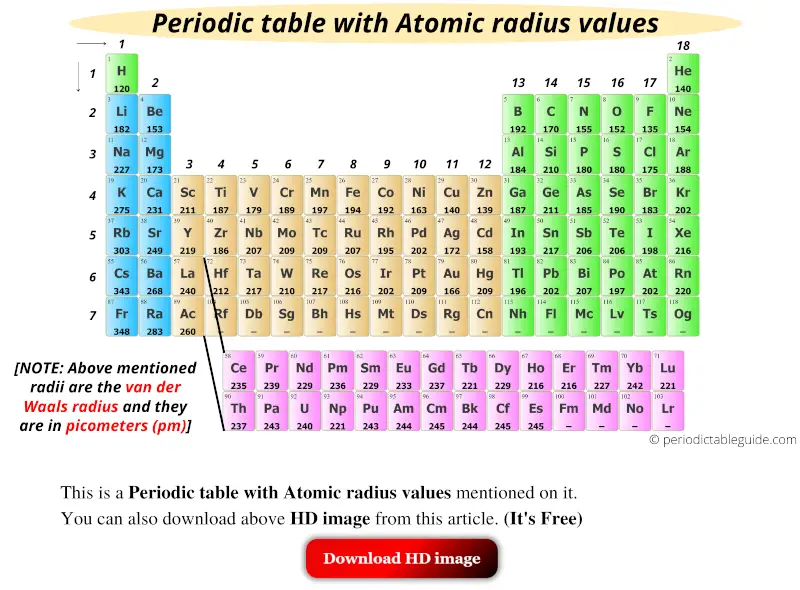
Get the Periodic table with Atomic radius values (Img+Chart)
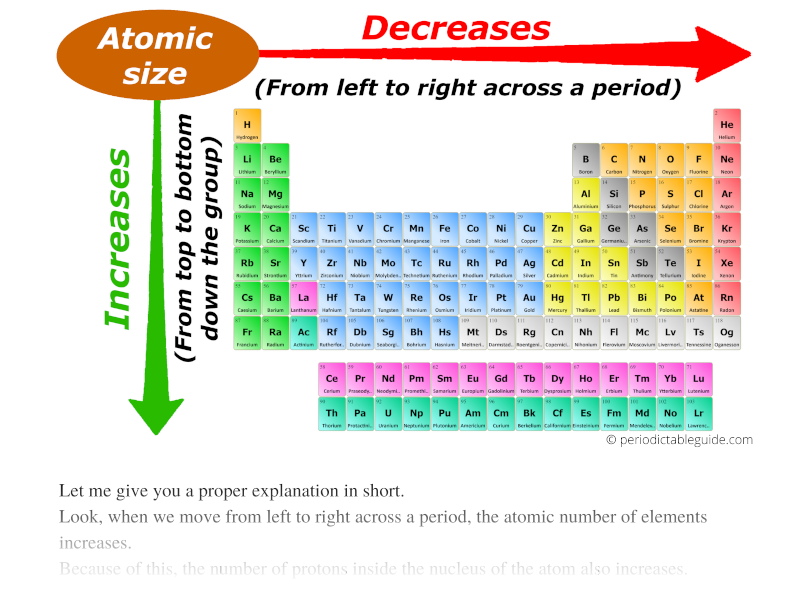
Atomic radius chart mindsstorm
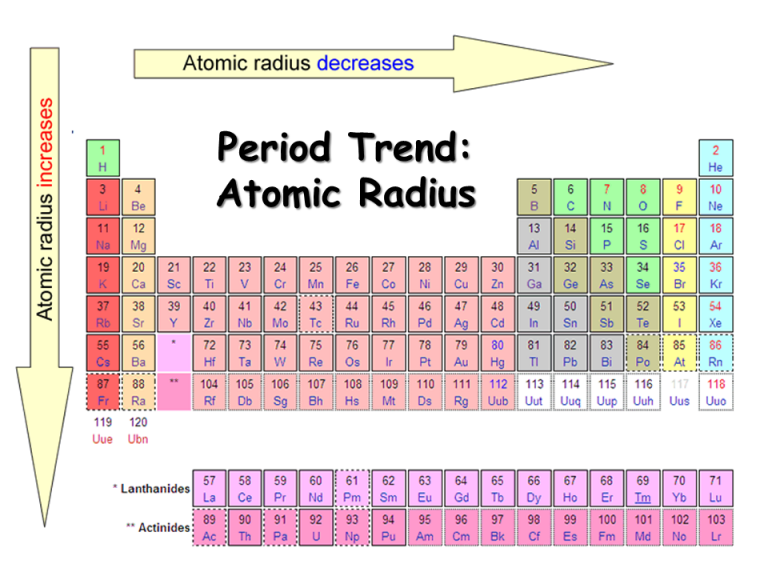
Atomic Radius NEETLab
The Above Atomic Radii Of The Elements Are The Van Der Waals Radius And They Are In Picometers (Pm).
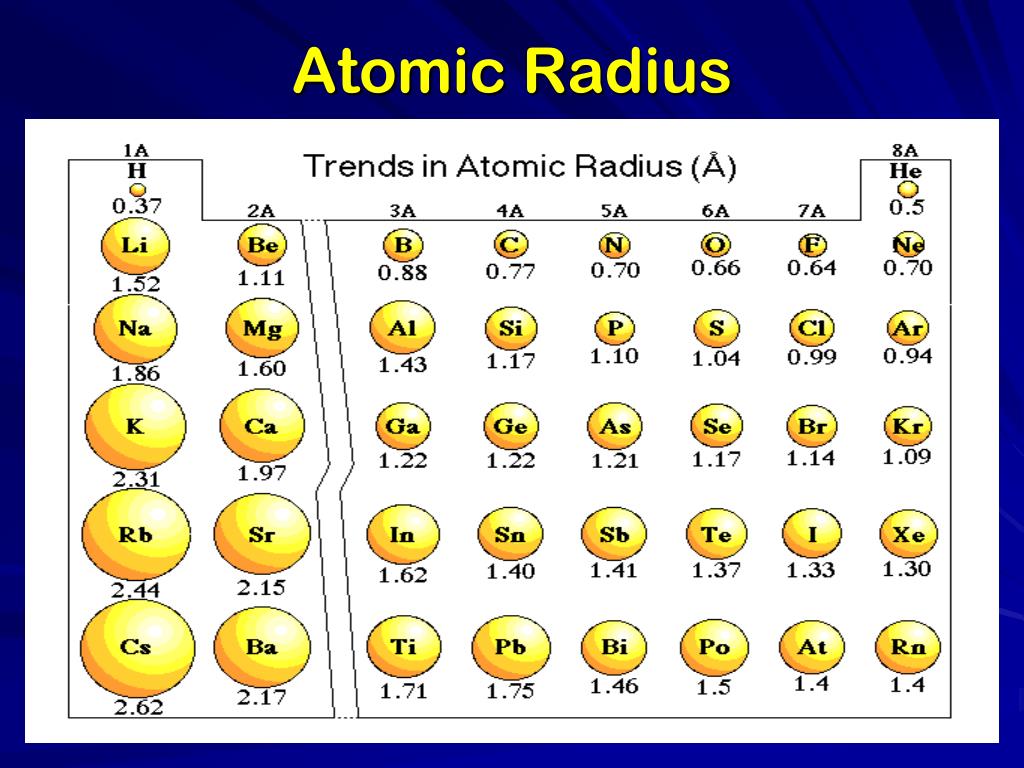
Web This Table Shows How The Atom Size, And Atomic Radius Values Change As You Move Horizontally And Vertically Across The Periodic Table.
The Largest Atom Is Cesium, While The Smallest Atom Is Helium.
Web Atomic Radius Trends On Periodic Table (Video) | Khan Academy.
Related Post: