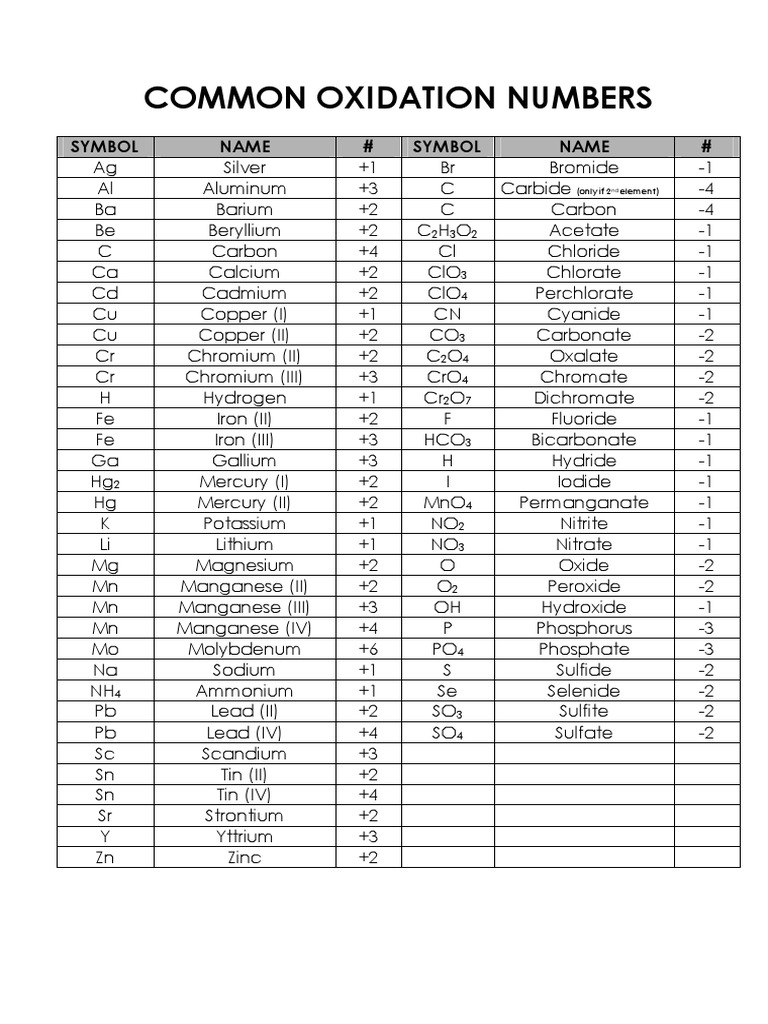
Chart Of Oxidation Number
Chart Of Oxidation Number - The most common oxidation states are in bold. Web determine what is the oxidizing and reducing agents in the following reaction. Web in simple words, the oxidation number is the number assigned to the components in a chemical combination. Web this color periodic table contains the number, symbol, name, atomic mass and oxidation states of each element. Oxidation number of an atom when an element has combined with the same element. You can't actually do that with vanadium, but you can with an element like sulphur. We can use oxidation numbers to keep track of where electrons are in a molecule, and how they move during a reaction. Elements have an oxidation state of zero, and atoms in ionic compounds are usually assigned a positive or negative oxidation state. First previous 1 next last. The oxidation number of hydrogen is +1 when it is combined with a nonmetal. In addition, the electron configurations of neutral atoms (oxidation state zero; Ch 4, nh 3, h 2 o, and hcl. The most common oxidation states are in bold text and predicted or unconfirmed states are in italics. Information of oxidation numbers of monoatomic ions in periodic table. Zn + 2h+ zn2+ +h2 zn + 2 h + zn 2 +. You can download the chart and the table above by clicking on either. The most common oxidation states are in bold text and predicted or unconfirmed states are in italics. Web enter the formula of a chemical compound to find the oxidation number of each element. The oxidation number is the total number of electrons that atoms in a molecule. To keep track of electrons in a. Each atom in a redox reaction is assigned an oxidation number to understand its ability to donate, accept, or share electrons. Web the following table lists the preferred (in bold), occasionally occurring, observed under certain conditions (in parentheses) and theoretically predicted oxidation states (in brackets) of the various chemical elements; Oxidation number of. Use uppercase for the first character in the. Each atom in a redox reaction is assigned an oxidation number to understand its ability to donate, accept, or share electrons. Web this printable periodic table contains the number, symbol, name, atomic mass and oxidation states of each element. Web this color periodic table contains the number, symbol, name, atomic mass and. If you're working out the oxidation states of the atoms in a reaction and you get one that's not on this chart, it's probably worth checking your work. The oxidation number, in simple terms, can be described as the number that is allocated to elements in a chemical combination. Web the chart below should help you to visualize the possible. Oxidation number of an atom when an element has combined with the same element. As the table shows, the presence of the other oxidation states varies, but follows some patterns. This is a list of all the known oxidation states of the chemical elements, excluding nonintegral values. Redox reactions are characterized by a transfer of electrons. The oxidation number is. We can use oxidation numbers to keep track of where electrons are in a molecule, and how they move during a reaction. Oxidation states of s block. Web the oxidation number is the positive or negative number of an atom that indicates the electrical charge the atom has if its compound consists of ions. As the table shows, the presence. Web list of oxidation states of the elements. Elements have an oxidation state of zero, and atoms in ionic compounds are usually assigned a positive or negative oxidation state. Web the oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction. This periodic table contains the oxidation numbers. Web showing 1 to 59 of 59 entries. We can use oxidation numbers to keep track of where electrons are in a molecule, and how they move during a reaction. Web what if you kept on adding electrons to the element? Web the oxidation number represents how many electrons an atom has gained or lost in a molecule. Web this. Web the following table lists the preferred (in bold), occasionally occurring, observed under certain conditions (in parentheses) and theoretically predicted oxidation states (in brackets) of the various chemical elements; In other words, the oxidation number gives the degree of oxidation (loss of electrons) or reduction (gain of electrons) of the atom in a compound. If you're working out the oxidation. Elements have an oxidation state of zero, and atoms in ionic compounds are usually assigned a positive or negative oxidation state. The oxidation number of hydrogen is +1 when it is combined with a nonmetal. Web the oxidation number is the positive or negative number of an atom that indicates the electrical charge the atom has if its compound consists of ions. As the table shows, the presence of the other oxidation states varies, but follows some patterns. Web the oxidation number of a monatomic (composed of one atom) ion is the same as the charge of the ion. In our water example, hydrogen is assigned an oxidation number of +1 because each individual hydrogen has lost one electron. Organic compounds and some covalent compounds do not have oxidation states assigned to the atoms in the. Web in simple words, the oxidation number is the number assigned to the components in a chemical combination. You can download the chart and the table above by clicking on either. Web list of oxidation states of the elements. Web this printable periodic table contains the number, symbol, name, atomic mass and oxidation states of each element. The oxidation number, in simple terms, can be described as the number that is allocated to elements in a chemical combination. Ch 4, nh 3, h 2 o, and hcl. Each atom in a redox reaction is assigned an oxidation number to understand its ability to donate, accept, or share electrons. The most common oxidation states are in bold text and predicted or unconfirmed states are in italics. Use uppercase for the first character in the.
Periodic Table Oxidation Chart

Labeled Periodic Table With Oxidation Numbers Periodic Table Timeline
Common Oxidation Numbers Chart

Oxidation Number Periodic table elements Definition, Rules (2023)
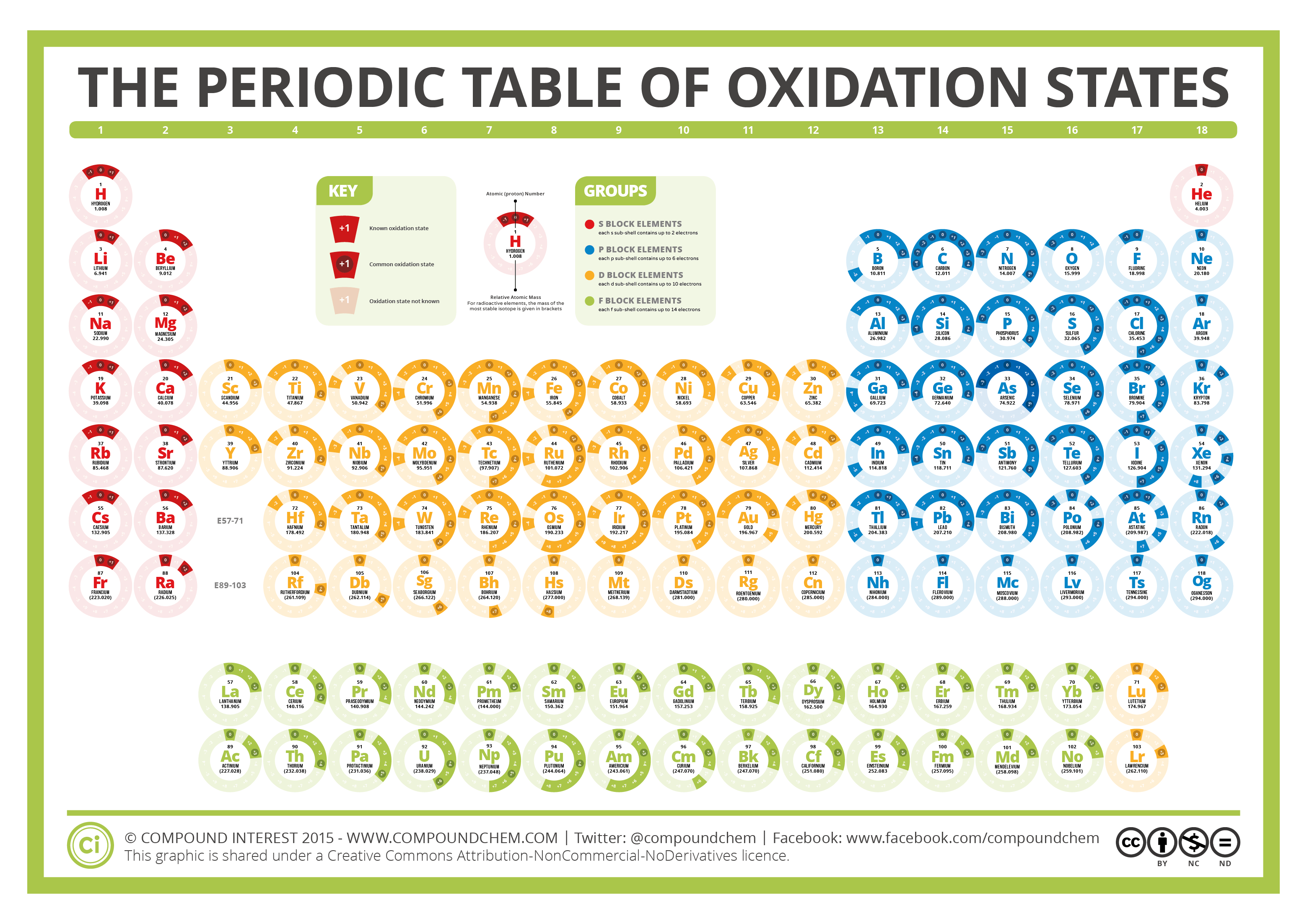
The Periodic Table of Oxidation States Compound Interest
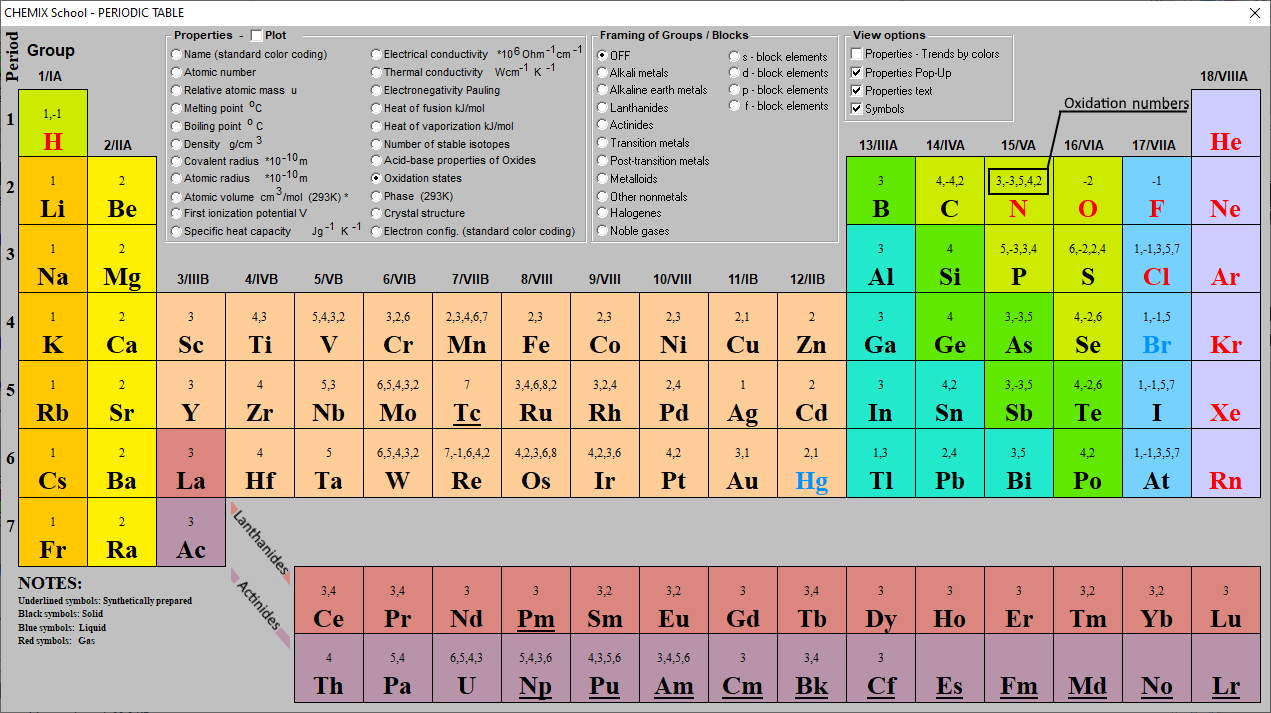
Oxidation Numbers Periodic Table Elements

Downloadable Periodic Table Oxidation States

oxidation states Archives Science Notes and Projects
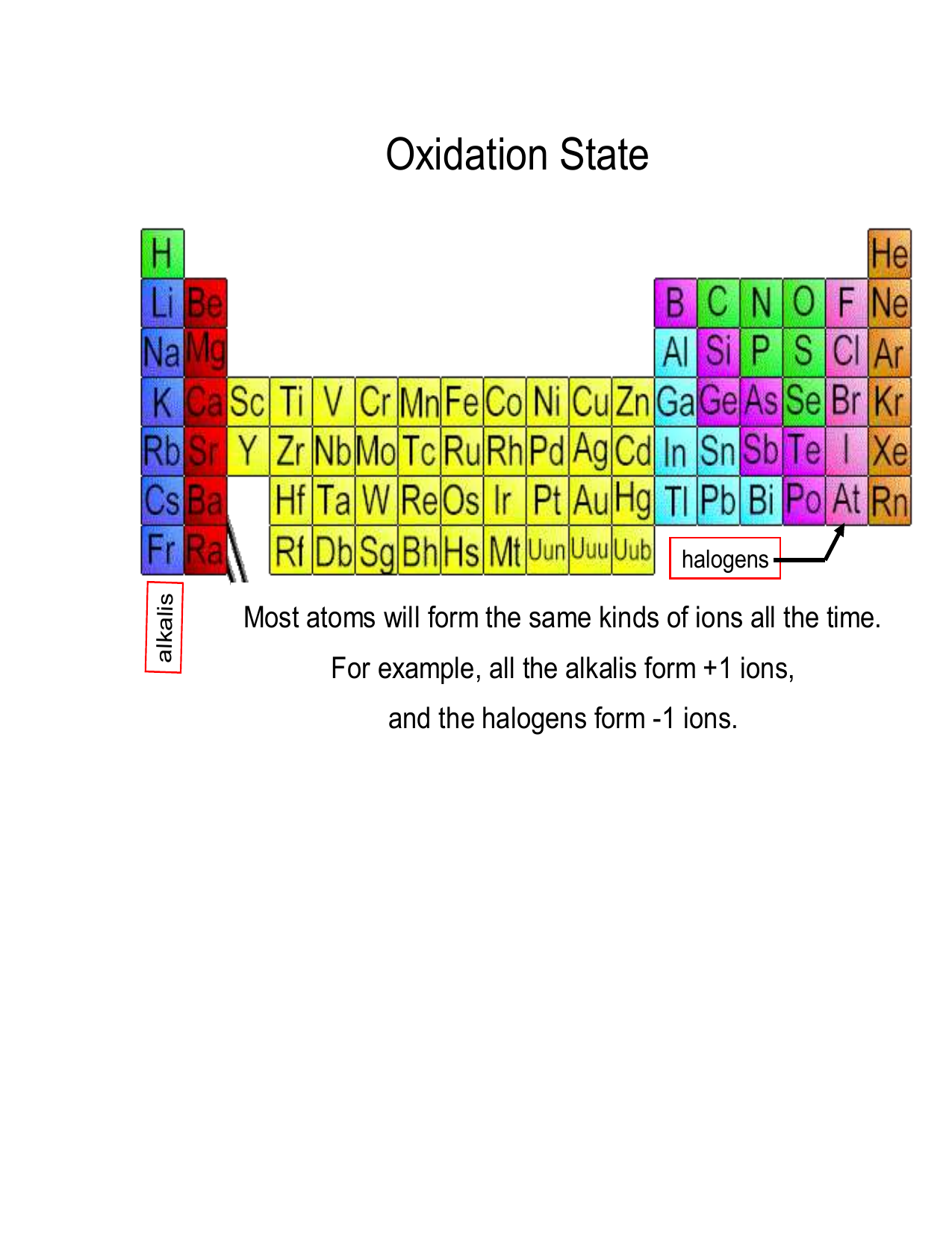
Oxidation Numbers
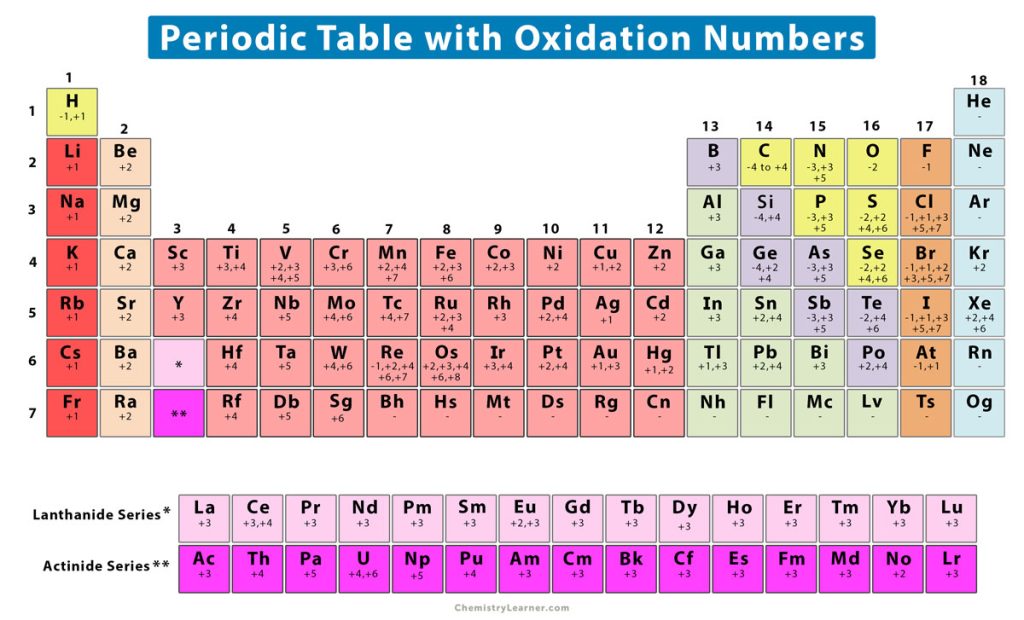
Oxidation Number (State) Definition, Rules, How to Find, and Examples
Web This Color Periodic Table Contains The Number, Symbol, Name, Atomic Mass And Oxidation States Of Each Element.
This Periodic Table Contains The Oxidation Numbers Of The Elements As Well As Element Numbers, Symbols, Names, And Atomic Weights.
Web An Oxidation Number Is A Number That Is Assigned To An Atom To Indicate Its State Of Oxidation Or Reduction During A Chemical Reaction.
This Table Is Based On Greenwood's,[1] With All Additions Noted.
Related Post: