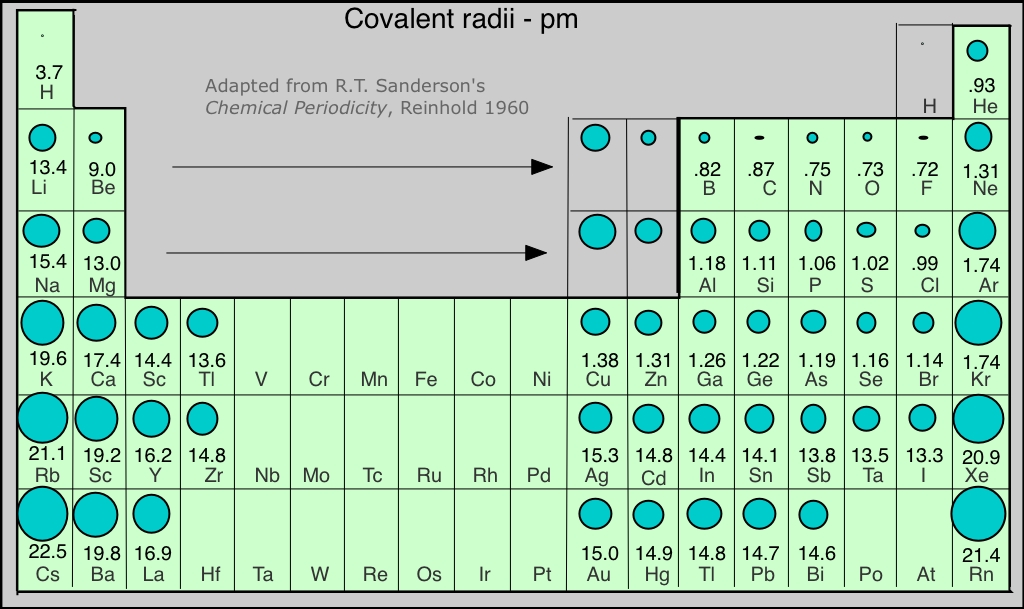
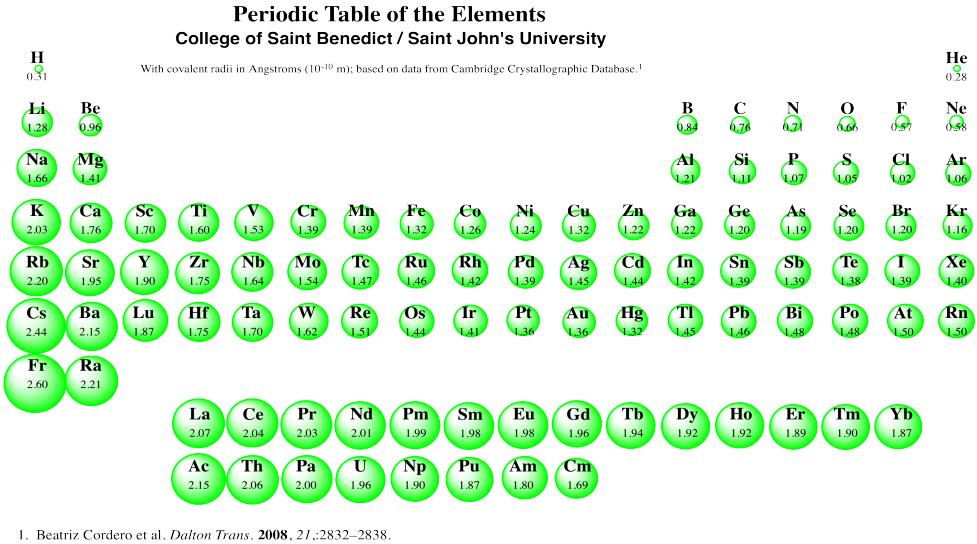
Covalent Radii Chart
Covalent Radii Chart - The differences between the two single. Web this is a set of covalent atomic radii (ref. Ionic radii are also available. It is usually measured either in picometres (pm) or angstroms (å), with 1 å = 100 pm. Web this figure shows radii (in angstroms) of atoms and ions of the first four periods of the periodic table. The covalent radius, rcov, is a measure of the size of an atom that forms part of one covalent bond. Web covalent radius increases as we move down a group because the n level (orbital size) increases. Moreover, different radii can be introduced for single, double and triple bonds (r1, r2 and r3 below), in a purely operational sense. 53 [citation needed] 120 or 110: Web the covalent radius is half the distance between two atoms that share a covalent bond. Web the covalent atomic radius (r cov) is half the internuclear distance in a molecule with two identical atoms bonded to each other, whereas the metallic atomic radius (r met) is. 1) deduced from crystallographic data for most of the elements with atomic numbers up to 96. These data show a well behaved periodic. Web covalent radius increases as we. Half of the single bond length between two similar atoms covalently bonded in a molecule is called the covalent radius. Ionic radii are also available. These data show a well behaved periodic. In principle, the sum of the two covalent radii should equal the covalent bond length between two atoms, r(ab) = r(a) + r(b). Ionic radii are also available. You will see a selection of chart images that show different aspects of covalent radius. Radii are measured from bonds to c, n or o. Web atomic radius of all the elements (complete chart) march 23, 2023. Web select from the following links to see visual periodicity representations for atomic radii, covalent radii, and van der waals radii. Ionic radii. These relationships are certainly not. Ionic radii are also available. A new set of covalent atomic radii has been deduced from crystallographic data for most of the elements with atomic numbers up to 96. Elements above 96 are assigned a covalent radius and uncertainty of none. Web atomic radius of all the elements (complete chart) march 23, 2023. In principle, the sum of the two covalent radii should equal the covalent bond length between two atoms, r(ab) = r(a) + r(b). Web covalent radius definition. Half of the single bond length between two similar atoms covalently bonded in a molecule is called the covalent radius. Radii are measured from bonds to c, n or o. Radii from each. Ionic radii are also available. Web these are he, ne, pm, at, rn, fr, ac, pa. Web this figure shows radii (in angstroms) of atoms and ions of the first four periods of the periodic table. Web this is a set of covalent atomic radii (ref. Web 119 rows covalent (single bond) covalent (triple bond) metallic 1: Web in theory, the sum of two covalent radii should equal the covalent bond length between two atoms, but in practice the length of the bond depends on the. These data show a well behaved periodic. Atomic radius of all the elements are mentioned in the chart below. In principle, the sum of the two covalent radii should equal the. It is usually measured either in picometres (pm) or angstroms (å), with 1 å = 100 pm. Moreover, different radii can be introduced for single, double and triple bonds (r1, r2 and r3 below), in a purely operational sense. In principle, the sum of the two covalent radii should equal the covalent bond length between two atoms, r(ab) = r(a). Radii are measured from bonds to c, n or o. Web covalent radius increases as we move down a group because the n level (orbital size) increases. Web the covalent atomic radius (r cov) is half the internuclear distance in a molecule with two identical atoms bonded to each other, whereas the metallic atomic radius (r met) is. Atomic radius. Web the values of table 3 show the covalent radii from the two recent original determinations by cordero et al.[1], and pyykkö and atsumi [2]. Web in this web page, you will find a covalent radius chart, a visual reference of charts. Web the covalent atomic radius (r cov) is half the internuclear distance in a molecule with two identical. Web this figure shows radii (in angstroms) of atoms and ions of the first four periods of the periodic table. Single bonds [1] single bonds [2] double bonds [2] triple bonds [2] 1: Atomic radius of all the elements are mentioned in the chart below. Web covalent radius increases as we move down a group because the n level (orbital size) increases. Ionic radii are also available. Covalent radius mostly decreases as we move left to right across a period. Web select from the following links to see visual periodicity representations for atomic radii, covalent radii, and van der waals radii. Web 120 rows covalent radii; The differences between the two single. Moreover, different radii can be introduced for single, double and triple bonds (r1, r2 and r3 below), in a purely operational sense. Radii are measured from bonds to c, n or o. Web in this web page, you will find a covalent radius chart, a visual reference of charts. The proposed radii show a well. 1) deduced from crystallographic data for most of the elements with atomic numbers up to 96. Usually, you see covalent radius in units of picometers (pm) or. Ionic radii are also available.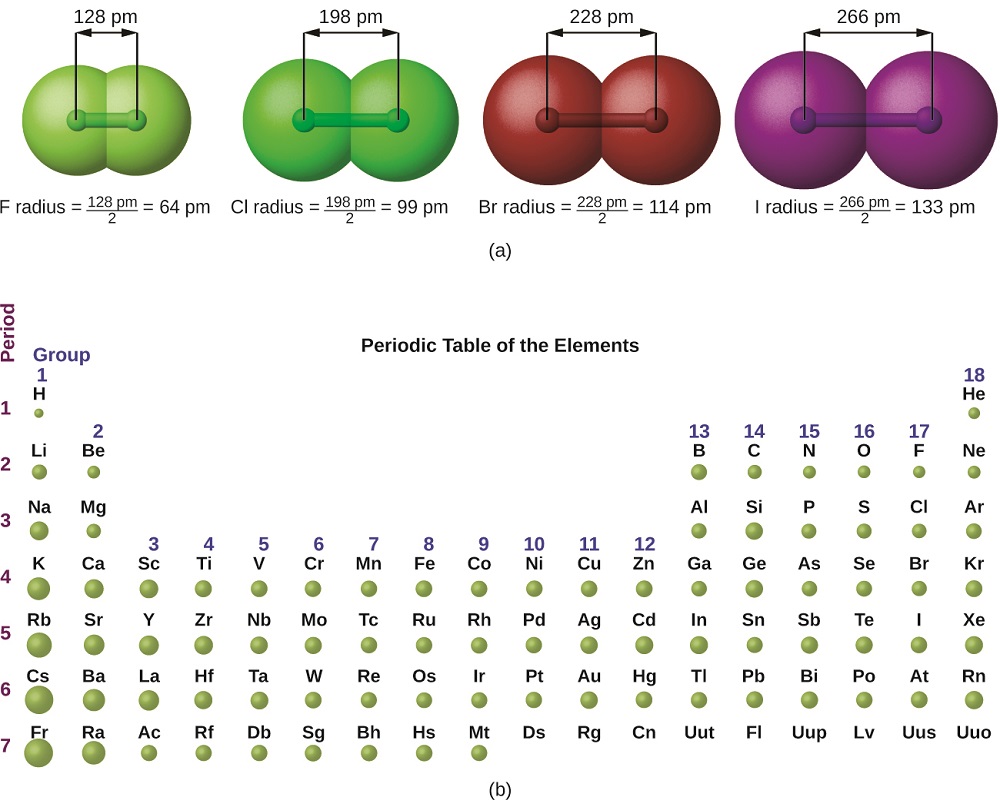
7.4 Electron Configurations, Valence Electrons, and the Periodic Table
What is the Atomic Radius? EnthuZiastic
5.7 Periodic Properties of the Elements Chemistry LibreTexts

Elements, Atomic Radii and the Periodic Radii

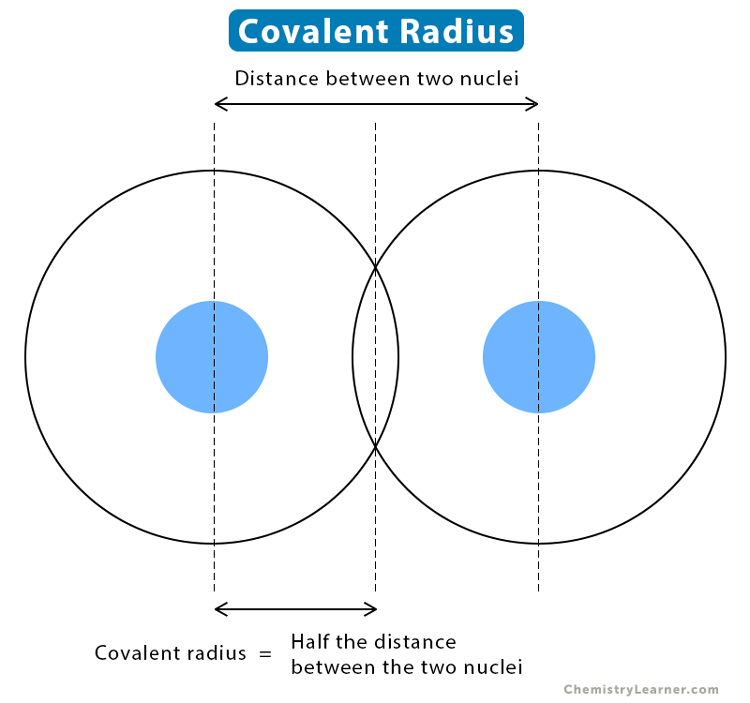
Covalent Radius Definition and Trend
Structure & Reactivity Appendix Periodic Radii
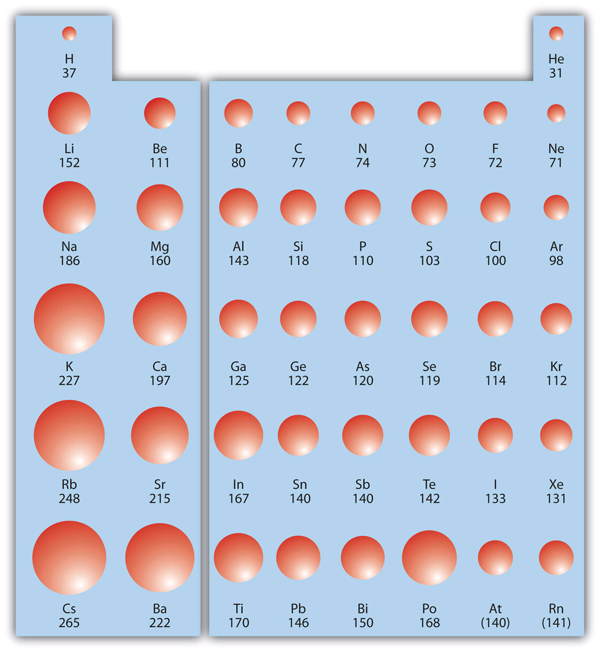
Periodic Trends Introductory Chemistry 1st Canadian Edition
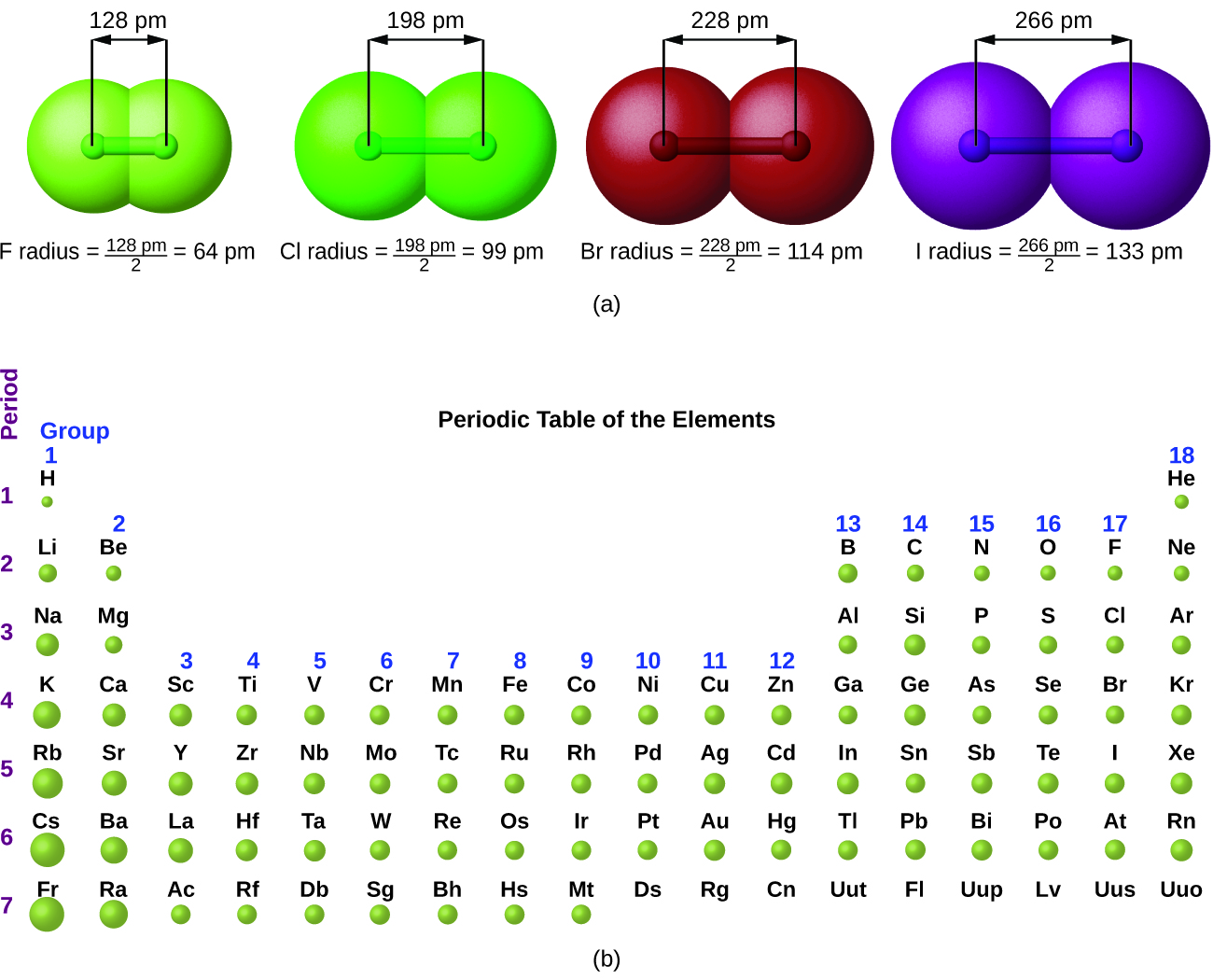
8.7 Periodic Trends and Variation of Properties General Chemistry
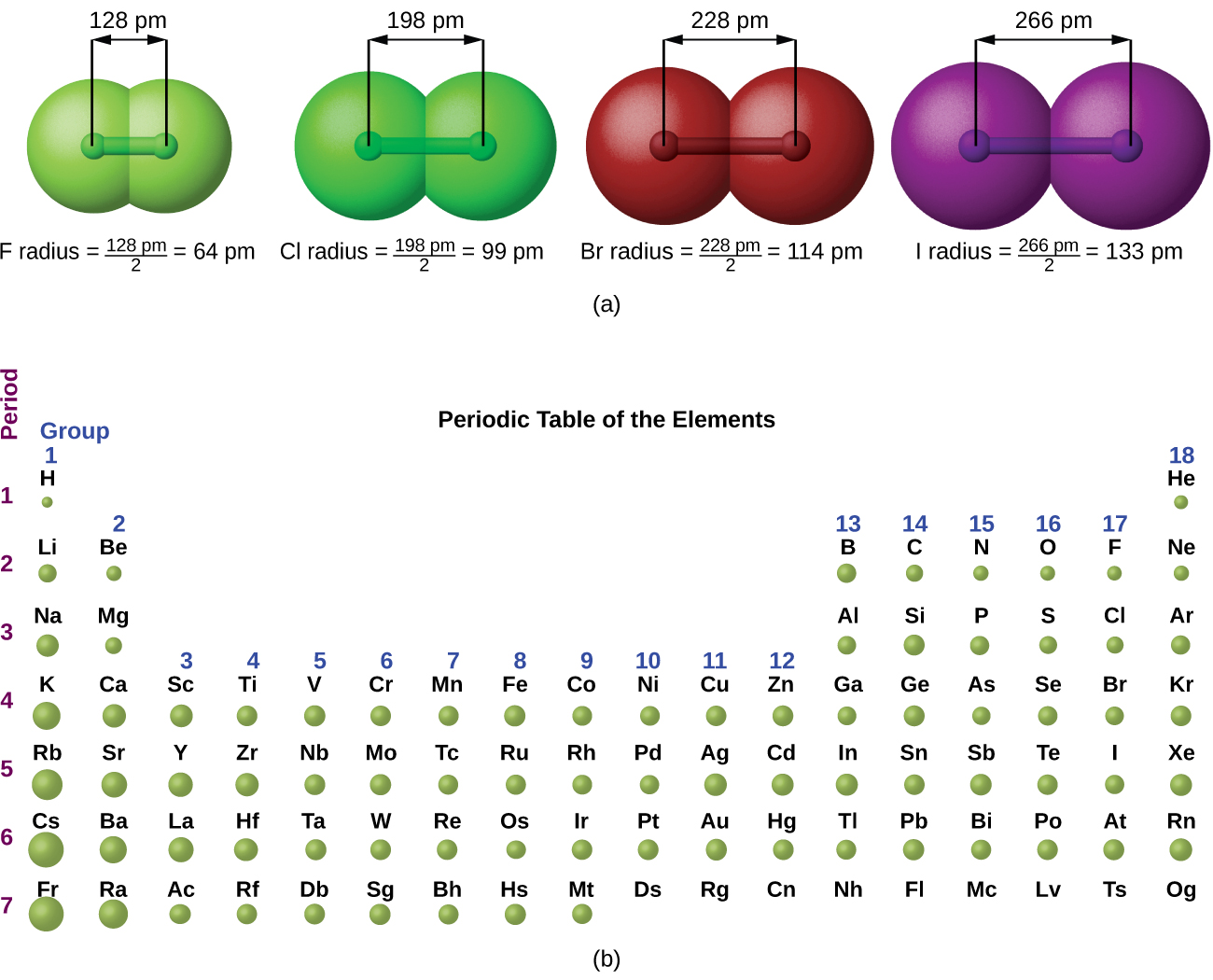
1.6 Periodic Variations in Element Properties Chemistry for

Covalent Radius Definition and Trend
In Principle, The Sum Of The Two Covalent Radii Should Equal The Covalent Bond Length Between Two Atoms, R(Ab) = R(A) + R(B).
Web Covalent Radius Definition.
Covalent Radii Are In Parentheses.
Radii From Each Element Are Listed From Largest To Smallest, Ionic Charge.
Related Post: