Draw The Lewis Structure For A Nitrogen Molecule
Draw The Lewis Structure For A Nitrogen Molecule - Figure out how many electrons the molecule must have, based on the number of valence electrons in each. The first step is to sketch the lewis structure of the n2 molecule, to add valence electrons around the two nitrogen atoms, and the final step is to combine the two nitrogen diatomic atoms to get the n2 lewis structure. Web lewis structures for covalent molecules: Draw the lewis structure for a nitrogen molecule, n2. Valence electrons that are not in a bond are shown as pairs of dots associated with individual atoms, lone pairs. Determine the total number of valence electrons the first step is to determine the total number of valence electrons for the molecule. Web to draw the n2 lewis structure, you can follow these steps: Web chemistry questions and answers. 2,5, it has five electrons in its outermost valence shell. In the case of n2, each nitrogen atom has five valence electrons, so the total number of valence electrons is 10. Web lewis structures for covalent molecules: You'll get a detailed solution from a subject matter expert that helps you learn core concepts. 2,5, it has five electrons in its outermost valence shell. Valence electrons that are not in a bond are shown as pairs of dots associated with individual atoms, lone pairs. It is produced in internal combustion engines when. Put the least electronegative atom in the center. Web steps of drawing lewis structure of n 2 molecule find total number of electrons of the valance shells of nitrogen atoms determine total electrons pairs existing as lone pairs and bonds find center atom selection mark lone pairs on atoms mark charges on atoms if there are charges. It is produced. Web chemistry questions and answers. As per the molecule n2, it has two atoms of nitrogen. In lewis structures, a single covalent bond is drawn as a pair of electron dots shared between two adjacent atoms, a bond pair. Web the nitrogen atom (group 15) has 5 valence electrons and each chlorine atom (group 17) has 7 valence electrons, for. Determine the total number of valence electrons in the molecule by adding the valence electrons of each nitrogen atom. The first step is to sketch the lewis structure of the n2 molecule, to add valence electrons around the two nitrogen atoms, and the final step is to combine the two nitrogen diatomic atoms to get the n2 lewis structure. Nitrogen. Web draw the lewis structure for a nitrogen (n2) molecule. Determine the total number of valence electrons in the molecule by adding the valence electrons of each nitrogen atom. It is produced in internal combustion engines when oxygen and nitrogen react at high temperatures. As per the molecule n2, it has two atoms of nitrogen. Find the total valence electrons. It is produced in internal combustion engines when oxygen and nitrogen react at high temperatures. As per the molecule n2, it has two atoms of nitrogen. Web the lewis dot structure of nitrogen consists of the nitrogen atom in the center with three dots surrounding it, representing its three valence electrons. Determine the total number of valence electrons the first. In the periodic table, nitrogen is placed in group 5 across period 2. It is produced in internal combustion engines when oxygen and nitrogen react at high temperatures. It is produced in internal combustion engines when oxygen and nitrogen react at high temperatures. Web drawing lewis structures for molecules with one central atom: Web the nitrogen atom (group 15) has. 2,5, it has five electrons in its outermost valence shell. Web the lewis dot structure of nitrogen consists of the nitrogen atom in the center with three dots surrounding it, representing its three valence electrons. Determine the total number of valence electrons the first step is to determine the total number of valence electrons for the molecule. The following procedure. Put the least electronegative atom in the center. Web the lewis structure of nitrogen atom can be drawn if one knows the number of valence electrons of nitrogen. The first step is to sketch the lewis structure of the n2 molecule, to add valence electrons around the two nitrogen atoms, and the final step is to combine the two nitrogen. Web to draw the n2 lewis structure, you can follow these steps: Valence electrons that are not in a bond are shown as pairs of dots associated with individual atoms, lone pairs. Web the lewis structure of nitrogen atom can be drawn if one knows the number of valence electrons of nitrogen. The electronic configuration of nitrogen is 1 #s^2#. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. It is produced in internal combustion engines when oxygen and nitrogen react at high temperatures. Nitrogen (n) is located in group 15 of the periodic table, so each nitrogen atom contributes five valence electrons. Web the lewis dot structure of nitrogen consists of the nitrogen atom in the center with three dots surrounding it, representing its three valence electrons. In the periodic table, nitrogen is placed in group 5 across period 2. Determine the total number of valence electrons in the molecule by adding the valence electrons of each nitrogen atom. The no2 lewis structure has a total of 17 valence electrons. Draw the lewis structure for a nitrogen molecule, n2. Determine the total number of valence electrons the first step is to determine the total number of valence electrons for the molecule. Thus, as per the electronic configuration of the element i.e. This problem has been solved! Valence electrons that are not in a bond are shown as pairs of dots associated with individual atoms, lone pairs. Web steps of drawing lewis structure of n 2 molecule find total number of electrons of the valance shells of nitrogen atoms determine total electrons pairs existing as lone pairs and bonds find center atom selection mark lone pairs on atoms mark charges on atoms if there are charges. In lewis structures, a single covalent bond is drawn as a pair of electron dots shared between two adjacent atoms, a bond pair. Put the least electronegative atom in the center. Nitrogen atom has 5 valence.Electron Dot Diagram For Nitrogen Wiring Diagram

Nitrogen Wikipedia
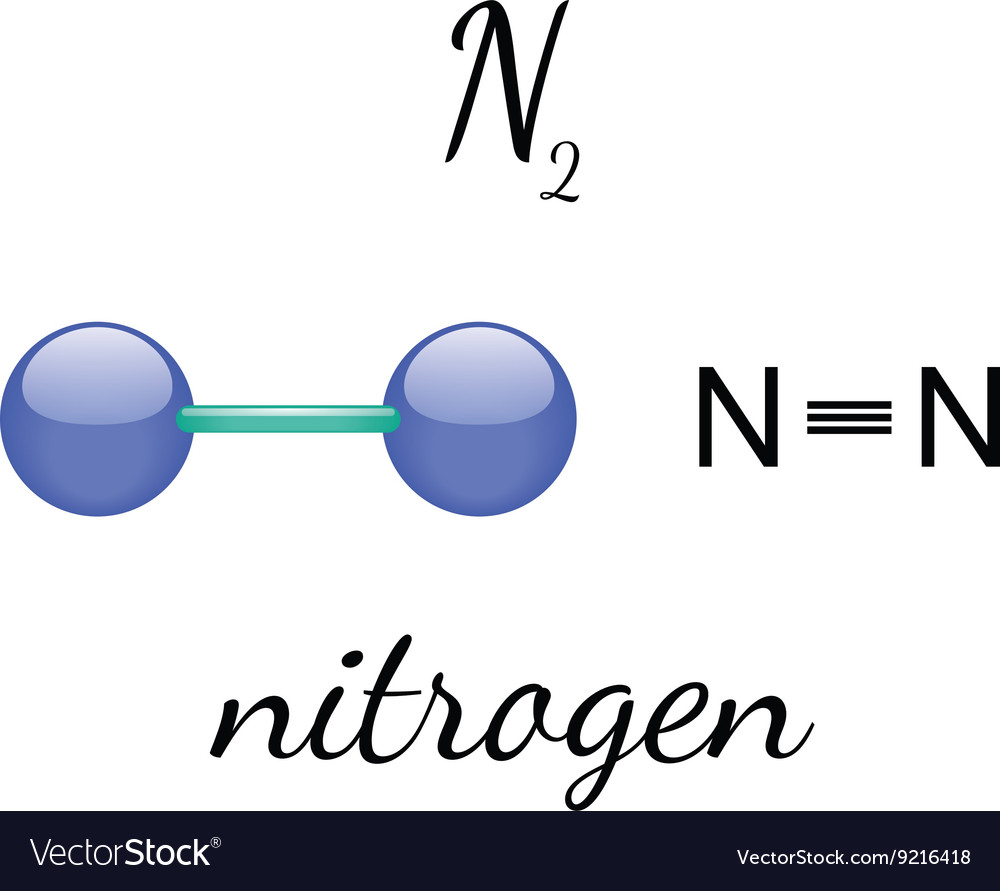
N2 nitrogen molecule Royalty Free Vector Image

Nitrogen Gas Lewis Dot Structure For Nitrogen Gas

【2 Steps】N2 Lewis StructureLewis Dot Structure for Nitrogen(N,N2

N2 Lewis Structure How to Draw the Lewis Structure for N2 Nitrogen Gas

Nitrogen Facts, Symbol, Discovery, Properties, Uses

How to Draw the Lewis Dot Structure for Diatomic Nitrogen (N2) YouTube
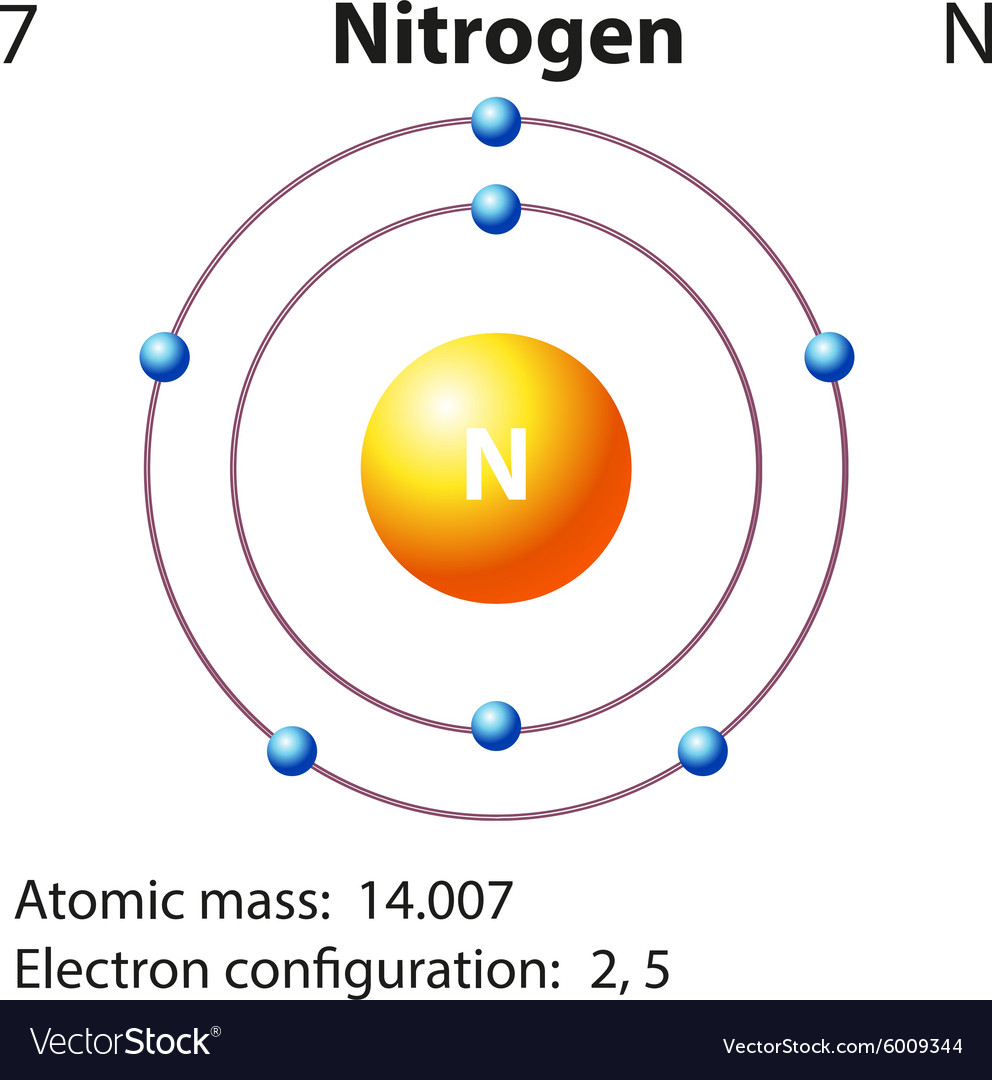
Diagram representation element nitrogen Royalty Free Vector
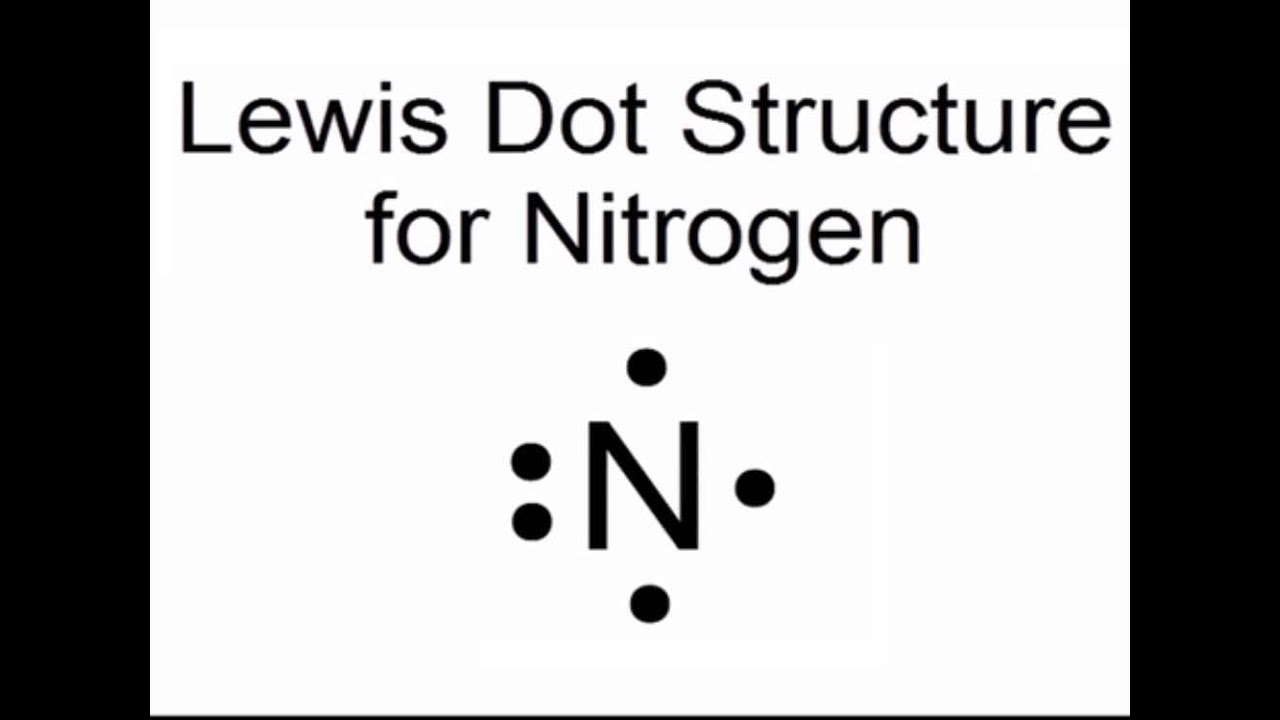
Lewis Dot Structure for Nitrogen Atom (N) YouTube
Web The Nitrogen Atom (Group 15) Has 5 Valence Electrons And Each Chlorine Atom (Group 17) Has 7 Valence Electrons, For A Total Of 26 Valence Electrons.
Web Chemistry Questions And Answers.
Web Drawing Lewis Structures For Molecules With One Central Atom:
Web The Nitrogen Atom (Group 15) Has 5 Valence Electrons And Each Chlorine Atom (Group 17) Has 7 Valence Electrons, For A Total Of 26 Valence Electrons.
Related Post:
