Draw The Lewis Structure For Sicl2Br2
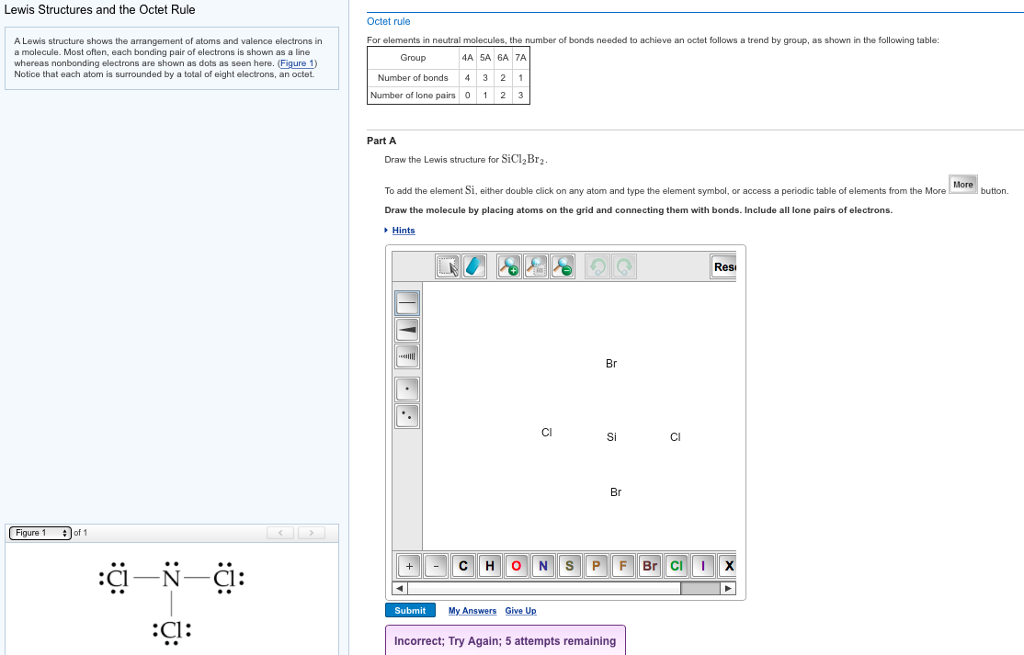
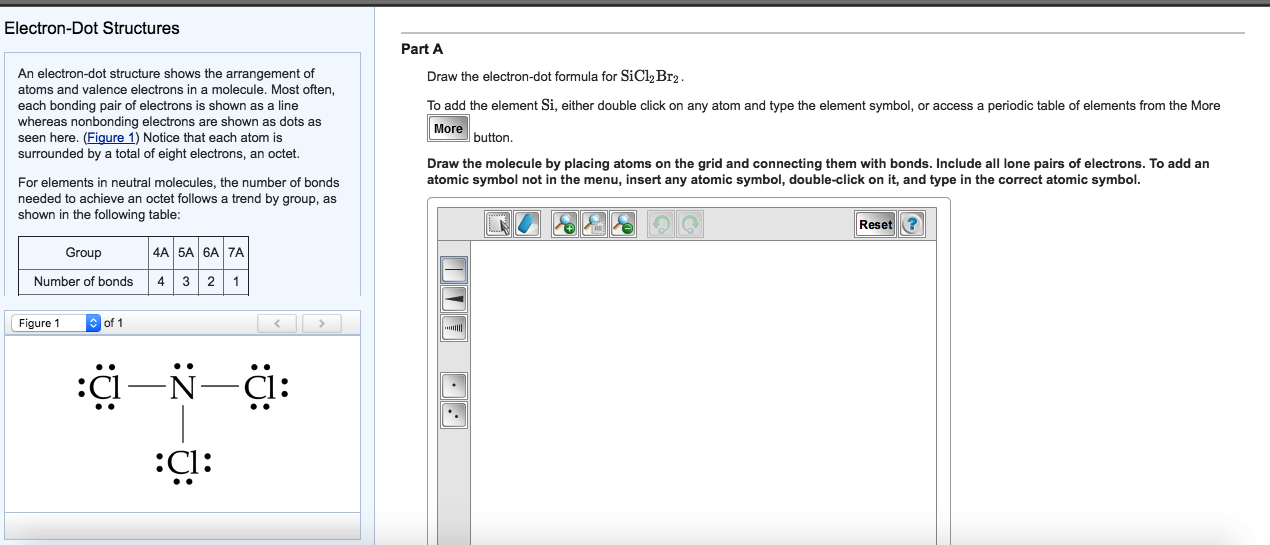
Draw The Lewis Structure For Sicl2Br2 - Web the lewis structure of sicl2br2 contains four single bonds, with silicon in the center, and two chlorines and two bromines on either side. Let us help you simplify your studying. As valance shell electrons play a significant role in this structural representation, counting of valance electrons should be completed firstly. Draw the molecule by placing atoms on the grid and connecting them with bonds. There are three lone pairs on each chlorine atom and each bromine atom, and the silicon atom does not have any lone pair. Draw the energy level diagram for c, h, o, n. Our videos will help you understand concepts, solve your homework, and do great on your exams. Include all lone pairs of electrons. Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons. To add the element si, either double click on any atom and type the element symbol, or access a periodic table of elements from the more button. Include all lone pairs of electrons. To add the element si, either double click on any atom and type the element symbol, or access a periodic table of elements from the more button.. Include all lone pairs of electrons. Web the lewis structure of sicl2br2 contains four single bonds, with silicon in the center, and two chlorines and two bromines on either side. It has four valence electrons. Here, the given molecule is sicl2br2. Include all lone pairs of electrons this problem has been solved! 4 (si) + 2(7) (cl) + 2(7) (br) = 32 step 2/5 Draw the molecule by placing atoms on the grid and connecting them with bonds. Web the lewis structure of sicl2br2 contains four single bonds, with silicon in the center, and two chlorines and two bromines on either side. Draw the molecule by placing atoms on the grid and. Draw the molecule by placing atoms on the grid and connecting them with bonds. Chlorine (cl) and bromine (br) are the surrounding atoms, each having seven valence electrons. Web drawing lewis structures for molecules with one central atom: Web draw the lewis structure for sicl2br2. Write down the electron configuration and draw lewis dot structure for each atom. Include all lone pairs of electrons. Web part a draw the lewis structure for sicl,br2. 4 (si) + 2(7) (cl) + 2(7) (br) = 32 step 2/5 And to help you understand the lewis structure. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. To add the element si, either double click on any atom and type the element symbol, or access a periodic table of elements from the more draw the molecule by placing atoms on the grid and connecting them with bonds. Web 6 steps to draw the lewis structure of sicl2br2 step #1: To add the element si, either double click. View available hint(s) 0 do q? Calculate the total number of valence electrons. Part a draw the lewis structure for sicl2br2. While selecting the center atom, always put the least electronegative atom at the. Web 6 steps to draw the lewis structure of sicl2br2 step #1: In sicl 2 br 2, silicon, chlorine (cl) and bromine (br) have 4,7 and 7 electrons respectively in their valance shell. Silicon (si) is the central atom in this molecule. While selecting the center atom, always put the least electronegative atom at the. Web to draw the lewis dot structure for sicl2br2, let's first determine the total number of valence. Web the following steps should be followed to draw the lewis structure of any molecule. Chlorine (cl) and bromine (br) are the surrounding atoms, each having seven valence electrons. Part a draw the lewis structure for sicl2br2. This widget gets the lewis structure of chemical compounds. Web part a draw the lewis structure for sicl,br2. Web steps to draw the lewis structure of sicl2br2. Web drawing lewis structures for molecules with one central atom: Draw the molecule by placing atoms on the grid and connecting them with bonds. Our videos will help you understand concepts, solve your homework, and do great on your exams. Count the total number of valence. 4 (si) + 2(7) (cl) + 2(7) (br) = 32 step 2/5 Let us help you simplify your studying. Web 1 2 3 4 5 6 7 8 9 0 1 2 3 4 5 6 7 8 9 share 226 views 1 year ago lewis structure sicl2br2 is a chemical formula for dibromo dichloro silane. Get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Web our videos prepare you to succeed in your college classes. To add the element si, either double click on any atom and type the element symbol, or access a periodic table of elements from the more button. Therefore, the total number of valence electrons in sicl2br2 is: View available hint(s) 0 do q? If you are having trouble with chemistry, organic, physics, calculus, or statistics, we got your back! Write down the electron configuration and draw lewis dot structure for each atom. Find the total valence electrons in sicl2br2 molecule in order to find the total valence electrons in a sicl2br2 molecule, first of all you should know the valence electrons present in silicon atom, chlorine atom as well as bromine atom. In sicl 2 br 2, silicon, chlorine (cl) and bromine (br) have 4,7 and 7 electrons respectively in their valance shell. Silicon (si) has 4 valence electrons, chlorine (cl) has 7 valence electrons, and bromine (br) has 7 valence electrons. There are three lone pairs on each chlorine atom and each bromine atom, and the silicon atom does not have any lone pair. Here, the given molecule is sicl2br2. Find more chemistry widgets in wolfram|alpha.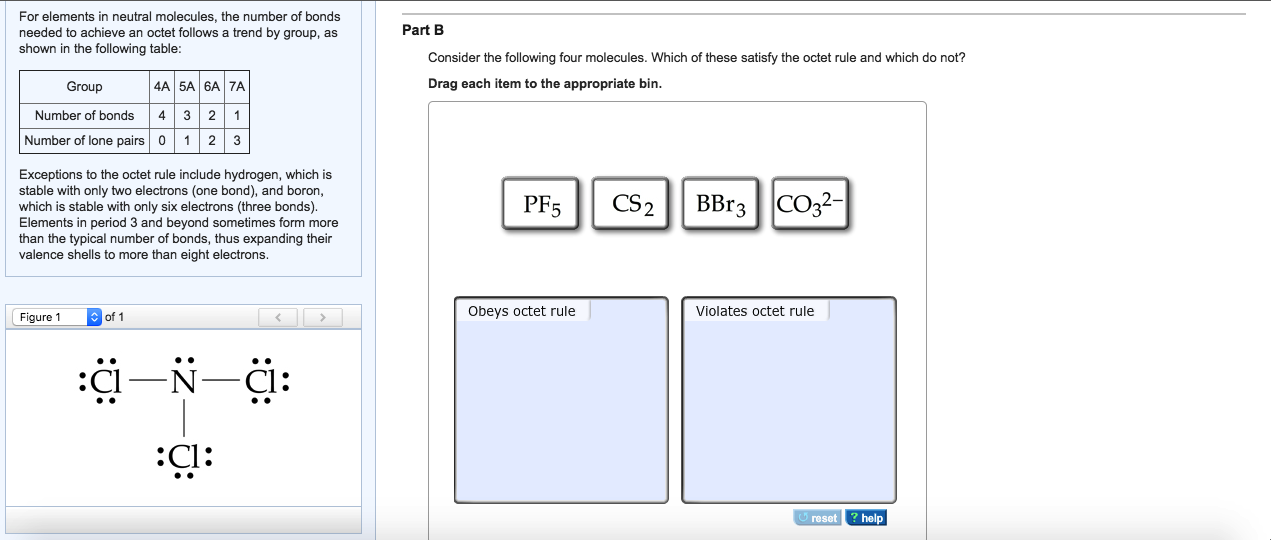
Sicl2br2 Lewis Structure How To Draw The Lewis Structure
lewis structure for sicl2br2
Draw The Lewis Structure For Sicl2br2
lewis structure for sicl2br2

SiCl2Br2 Lewis Structure How to Draw the Lewis Structure for SiCl2Br2
lewis structure for sicl2br2
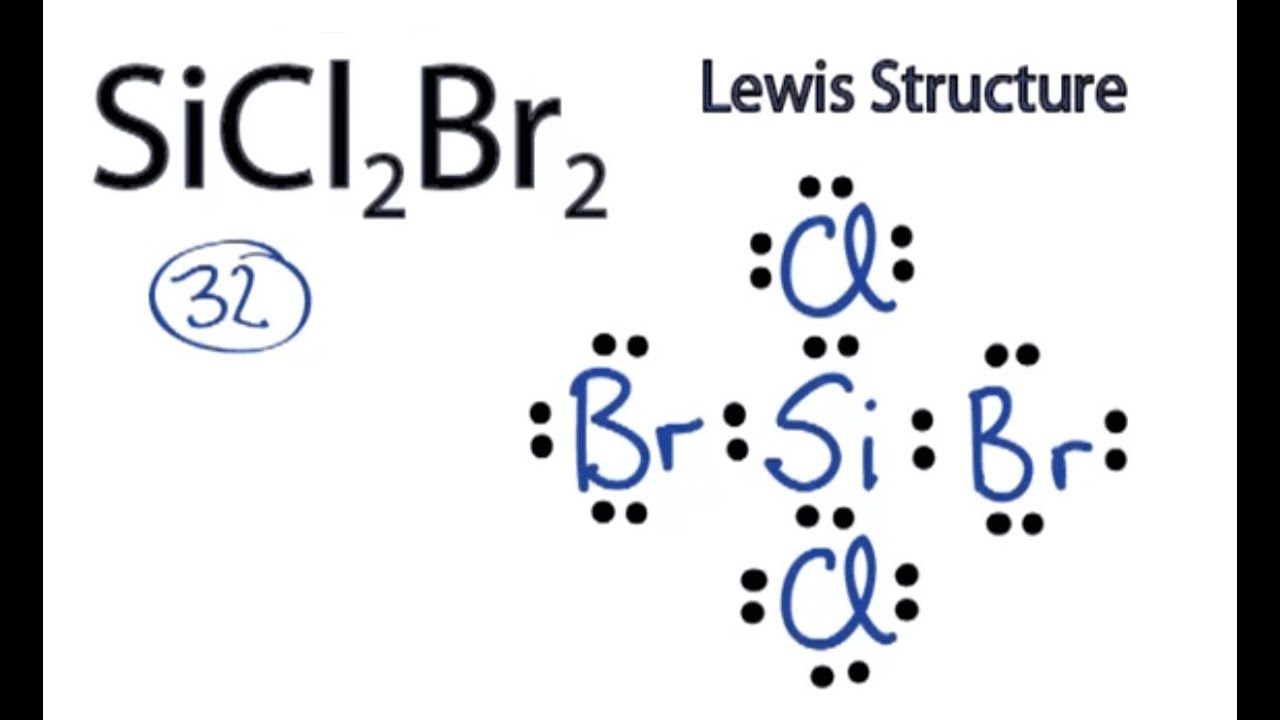
SiCl2Br2 Lewis Structure How to Draw the Lewis Structure for SiCl2Br2
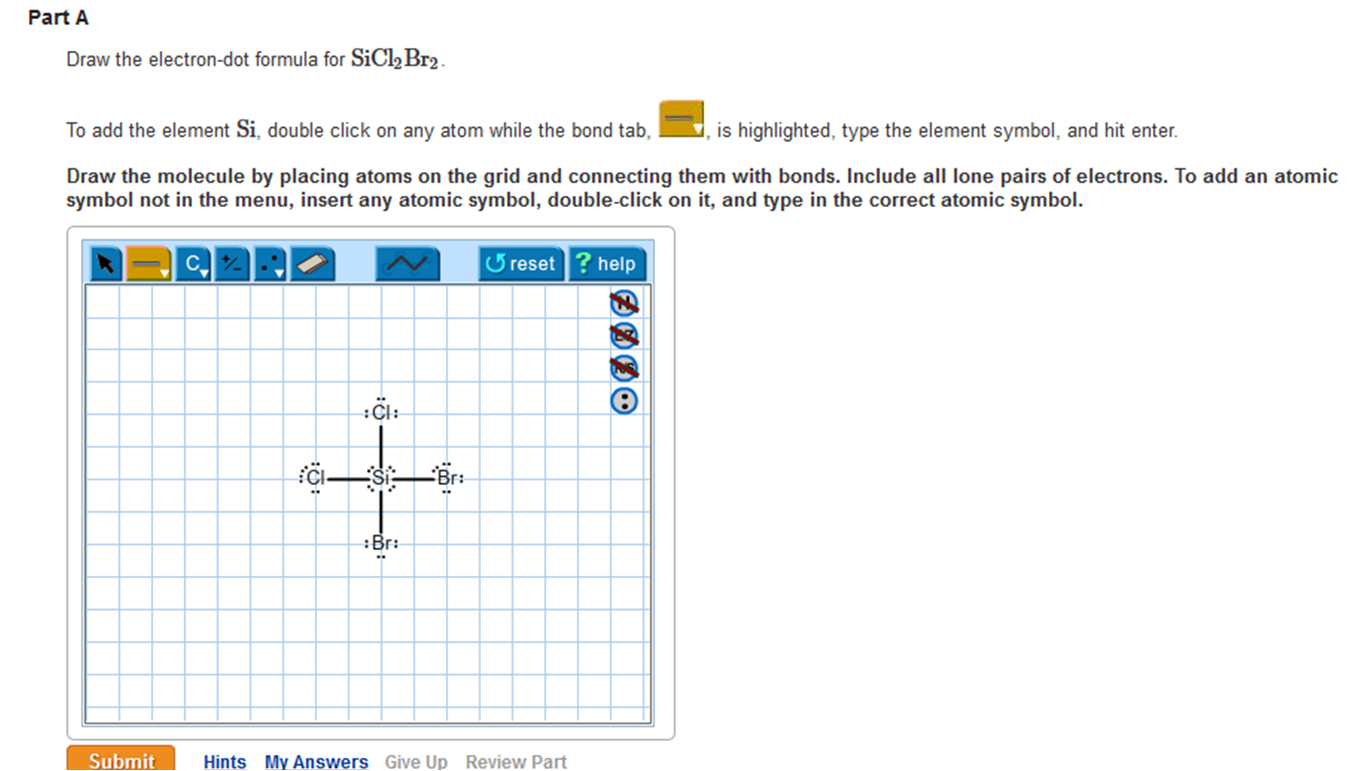
Solved Draw the electrondot formula for SiCl2Br2. This is
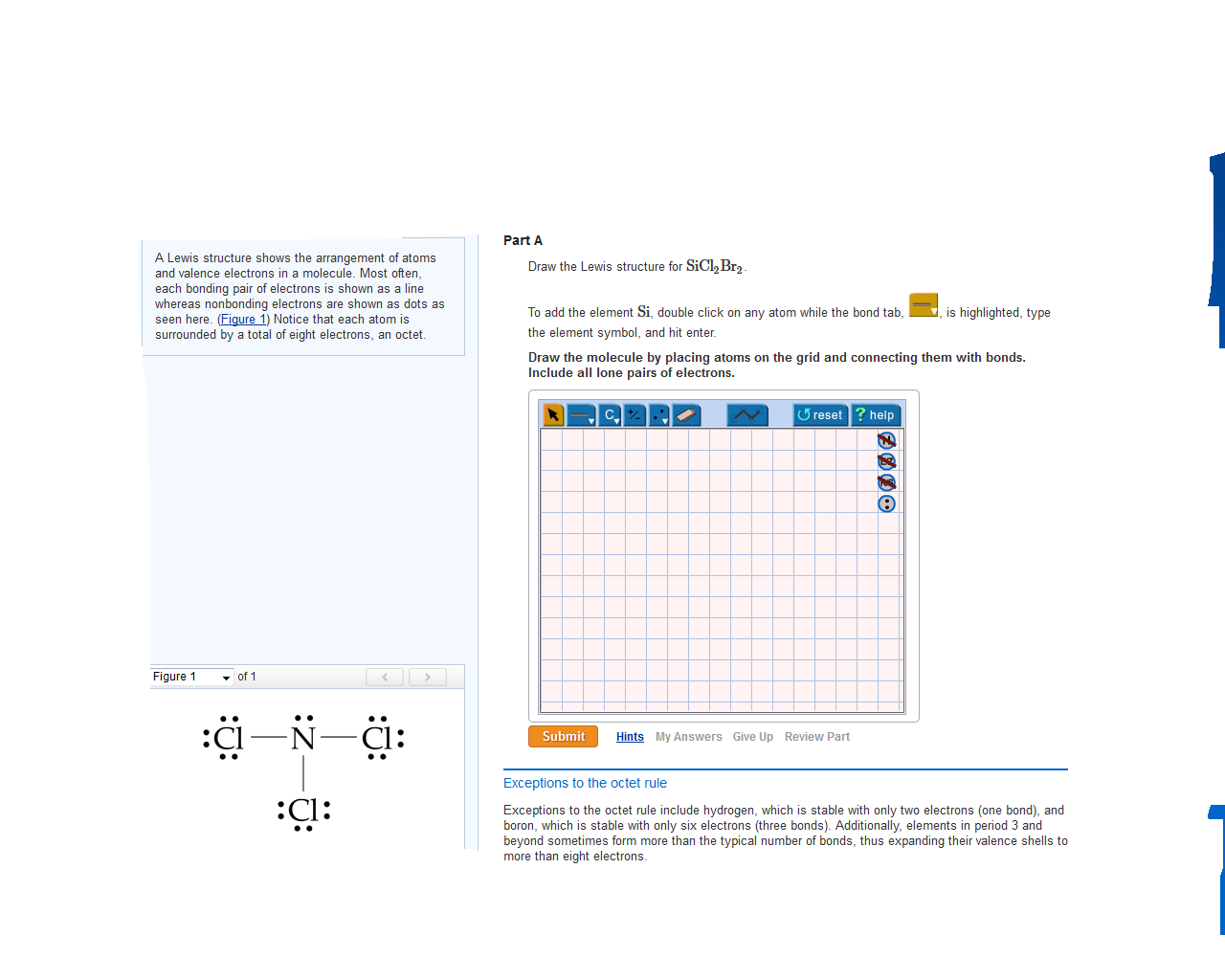
Sicl2br2 Lewis Structure How To Draw The Lewis Structure

Draw the Lewis Structure for Sicl2br2.
Silicon (Si) Is The Central Atom In This Molecule.
Ed Lone Pairs Of Electrons.
To Add The Element Si, Either Double Click On Any Atom And Type The Element Symbol, Or Access A Periodic Table Of Elements From The More Draw The Molecule By Placing Atoms On The Grid And Connecting Them With Bonds.
Web The Lewis Structure Of Sicl2Br2 Contains Four Single Bonds, With Silicon In The Center, And Two Chlorines And Two Bromines On Either Side.
Related Post: