Enthalpy Entropy Chart
Enthalpy Entropy Chart - Enthalpy is the heat content of a system. The enthalpy change of a reaction is roughly equivalent to the amount of energy lost or gained during the reaction. Web the diagram below can be used to determine enthalpy versus entropy of water and steam. It is denoted by the symbol e. It is a state function. In general, it is a relationship between enthalpy (measure of the energy of a thermodynamic system), air temperature, and moisture content. The mollier diagram is a graph used in thermodynamics to visualize the relationships between temperature, pressure, specific volume, enthalpy, and entropy of a substance. Denoted as δs δ s, the change of entropy suggests that time itself is asymmetric with respect to order of an isolated system, meaning: The standard enthalpy of formation of hcl(g) is −92.3 kj/mol. A system will become more disordered, as time increases. The mollier diagram is a graph used in thermodynamics to visualize the relationships between temperature, pressure, specific volume, enthalpy, and entropy of a substance. A system will become more disordered, as time increases. Enthalpy is the heat content of a system. A reaction is favored if the enthalpy of the. Denoted as δs δ s, the change of entropy suggests. In general, it is a relationship between enthalpy (measure of the energy of a thermodynamic system), air temperature, and moisture content. Web what is the enthalpy change for the reaction of 1 mole of h 2 (g) with 1 mole of cl 2 (g) if both the reactants and products are at standard state conditions? It is a state function.. These lines extend at an angle from the saturated vapor line. Web the diagram below can be used to determine enthalpy versus entropy of water and steam. The state of the compound is specified by the following symbols: The standard enthalpy of formation of hcl(g) is −92.3 kj/mol. For the reaction \[\ce{h2}(g)+\ce{cl2}(g) \ce{2hcl}(g)\hspace{20px}δh^\circ_{298}=\mathrm{−184.6\:kj. Denoted as δs δ s, the change of entropy suggests that time itself is asymmetric with respect to order of an isolated system, meaning: Web the thermodynamic arrow of time (entropy) is the measurement of disorder within a system. Web for now, we will just look at enthalpy. The standard enthalpy of formation of hcl(g) is −92.3 kj/mol. Web a. Most engineers understand the role units play in definition and verification of the engineering concepts, principles, equations and. Web for now, we will just look at enthalpy. Web the figures and tables below shows how water enthalpy and entropy changes with temperature (°c and °f) at water saturation pressure (which for practicle use, gives the same result as atmospheric pressure. Denoted as δs δ s, the change of entropy suggests that time itself is asymmetric with respect to order of an isolated system, meaning: These lines extend at an angle from the saturated vapor line. In general, it is a relationship between enthalpy (measure of the energy of a thermodynamic system), air temperature, and moisture content. Web the diagram below. It is a state function. The state of the compound is specified by the following symbols: Enthalpy is the heat content of a system. Web the diagram below can be used to determine enthalpy versus entropy of water and steam. These values are especially useful for computing or predicting enthalpy changes for chemical reactions that are impractical or. It is denoted by the symbol e. Web the diagram below can be used to determine enthalpy versus entropy of water and steam. Web the figures and tables below shows how water enthalpy and entropy changes with temperature (°c and °f) at water saturation pressure (which for practicle use, gives the same result as atmospheric pressure at temperatures < 100. Most engineers understand the role units play in definition and verification of the engineering concepts, principles, equations and. Web the standard free energy of formation of a compound can be calculated from the standard enthalpy of formation (δh ∘ f) and the standard entropy of formation (δs ∘ f) using the definition of free energy: The mollier diagram is a. Major players in developing the second law. The enthalpy change of a reaction is roughly equivalent to the amount of energy lost or gained during the reaction. Web the diagram below can be used to determine enthalpy versus entropy of water and steam. A reaction is favored if the enthalpy of the. Web the thermodynamic arrow of time (entropy) is. These values are especially useful for computing or predicting enthalpy changes for chemical reactions that are impractical or. For the reaction \[\ce{h2}(g)+\ce{cl2}(g) \ce{2hcl}(g)\hspace{20px}δh^\circ_{298}=\mathrm{−184.6\:kj. In general, it is a relationship between enthalpy (measure of the energy of a thermodynamic system), air temperature, and moisture content. The mollier diagram is a graph used in thermodynamics to visualize the relationships between temperature, pressure, specific volume, enthalpy, and entropy of a substance. A system will become more disordered, as time increases. It is denoted by the symbol e. Most engineers understand the role units play in definition and verification of the engineering concepts, principles, equations and. It is a state function. Web the thermodynamic arrow of time (entropy) is the measurement of disorder within a system. Web definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for standard enthalpy and gibbs free energy of formation, as well as standard entropy and molar heat capacity, of 370 inorganic compounds. Web for now, we will just look at enthalpy. Enthalpy is defined as the sum of the internal energy of a system and the product of its pressure and volume. Web what is the enthalpy change for the reaction of 1 mole of h 2 (g) with 1 mole of cl 2 (g) if both the reactants and products are at standard state conditions? Web the standard free energy of formation of a compound can be calculated from the standard enthalpy of formation (δh ∘ f) and the standard entropy of formation (δs ∘ f) using the definition of free energy: The standard enthalpy of formation of hcl(g) is −92.3 kj/mol. Denoted as δs δ s, the change of entropy suggests that time itself is asymmetric with respect to order of an isolated system, meaning: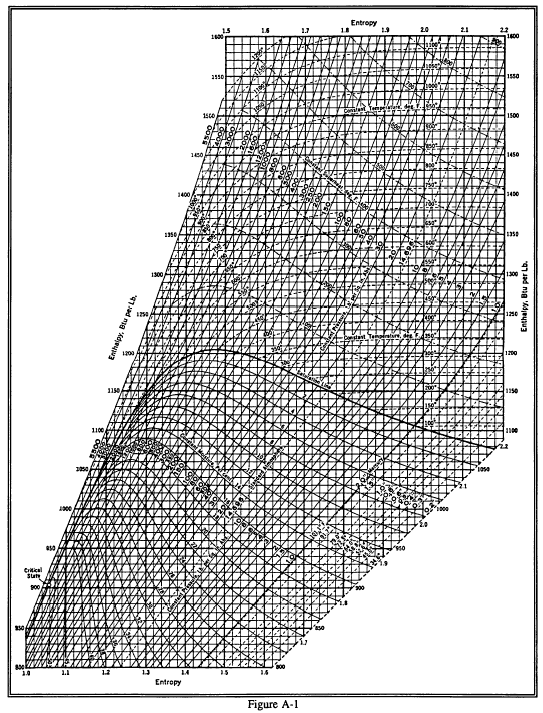
Enthalpy Entropy (hs) or Mollier Diagram Engineers Edge www

Enthalpy And Entropy Chart
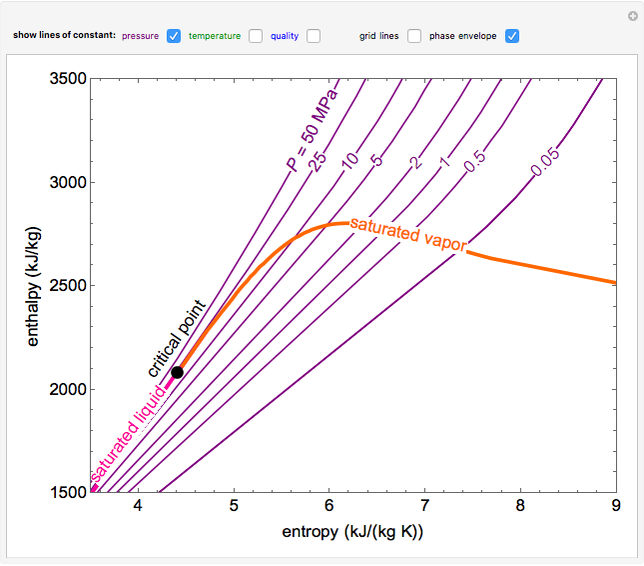
enthalpyentropydiagramforwater LearnChemE
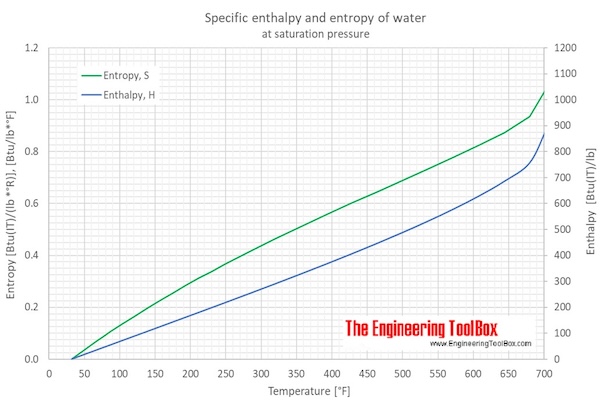
Water Enthalpy and Entropy vs. Temperature
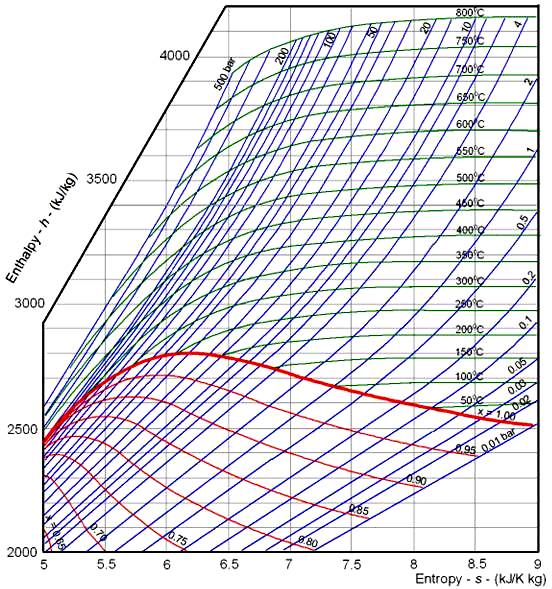
Enthalpy And Entropy Pdf flavilen
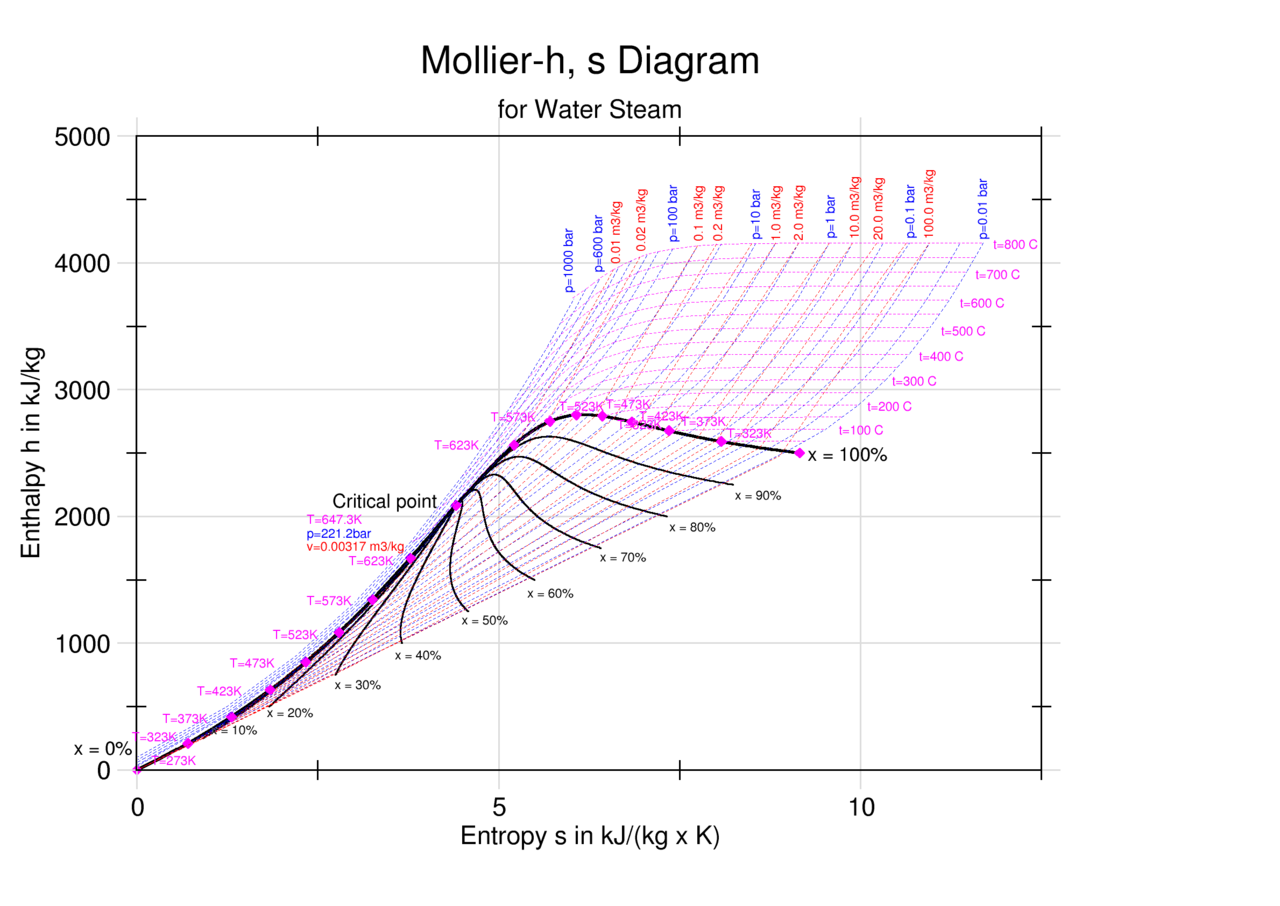
Enthalpy Entropy (hs) or Mollier Diagram
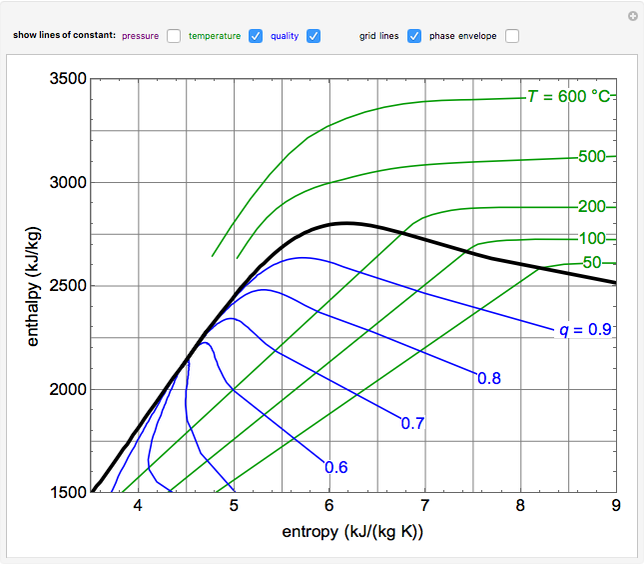
EnthalpyEntropy Diagram for Water Wolfram Demonstrations Project

Entropy And Enthalpy Chart
Enthalpy And Entropy Chart
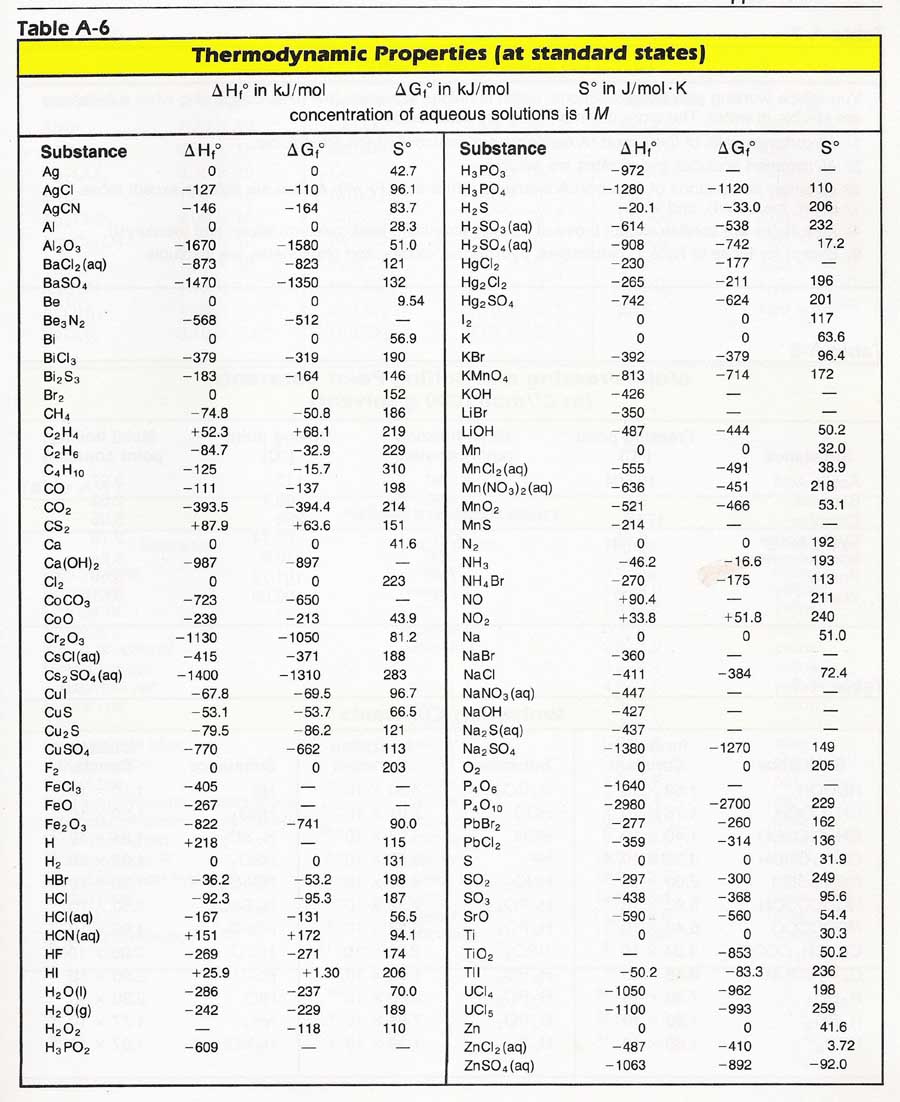
Entropy Table
Units Used To Express Enthalpy Are Calorie, Btu, Or Joules.
These Lines Extend At An Angle From The Saturated Vapor Line.
A Reaction Is Favored If The Enthalpy Of The.
Web A Standard Enthalpy Of Formation Is An Enthalpy Change For A Reaction In Which Exactly 1 Mole Of A Pure Substance Is Formed From Free Elements In Their Most Stable States Under Standard State Conditions.
Related Post: