Galvanic Chart For Metals
Galvanic Chart For Metals - Though the order of metals in a galvanic series remains the same in most conducting solutions, some. The chart can be used to determine the likelihood of a galvanic reaction, and galvanic corrosion or bimetallic corrosion, between two different metals in a seawater environment. Water) is necessary to promote galvanic corrosion. The relative position of two metals on such a series gives a good indication of. So, for example, choosing zinc on zinc would have the lowest risk for corrosion. Web below is a galvanic reaction chart for dissimilar metals. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. In this article, we will discuss what is galvanic corrosion, its applications, and how to prevent it using a galvanic corrosion chart? Web typically, the presence of an electrolyte (eg. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. Water) is necessary to promote galvanic corrosion. Web fastener material selection based on the galvanic series of metals to minimize galvanic corrosion, select fasteners based on their material compatibility with the substrates. Web the table below is the galvanic series of metals, alloys and graphite in seawater (most noble at top) in flowing seawater, at ‘normal’ temperature. Web below is. Web view this chart of galvanic compatibility. Web typically, the presence of an electrolyte (eg. Water) is necessary to promote galvanic corrosion. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. Web the galvanic series chart below shows metals and their electrochemical voltage range. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). First there must be two electrochemically dissimilar metals present. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. Maximum recommended voltage difference is 0,2v: The. Web fastener material selection based on the galvanic series of metals to minimize galvanic corrosion, select fasteners based on their material compatibility with the substrates. Electric current flows from plus to minus. The position of zinc on the galvanic series, above most other metals, means that it will corrode preferentially if it contacts any of these metals and moisture is. Web because of this, it’s important to understand which metal combinations could create the most risk potential for galvanic corrosion. Electric current flows from plus to minus. Web however, you can completely avoid galvanic corrosion by choosing matching metal anchors. Web fastener material selection based on the galvanic series of metals to minimize galvanic corrosion, select fasteners based on their. Web all metals can be classified into a galvanic series representing the electrical potential they develop in a given electrolyte against a standard reference electrode. Water) is necessary to promote galvanic corrosion. So, for example, choosing zinc on zinc would have the lowest risk for corrosion. Web below is a galvanic reaction chart for dissimilar metals. Web some metals like. Web below is a galvanic reaction chart for dissimilar metals. To use the chart, align the metal to be assessed (for the risk of corrosion) in the left column with the contact metal listed in the. Water) is necessary to promote galvanic corrosion. Web all metals can be classified into a galvanic series representing the electrical potential they develop in. Web a chart depicting the galvanic series for some common metals in a frequently encountered conducting solution, seawater, is included in figure 1. Web typically, the presence of an electrolyte (eg. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). So, for example, choosing zinc on zinc would have the. In this article, we will discuss what is galvanic corrosion, its applications, and how to prevent it using a galvanic corrosion chart? Web this chart will help you to determine which metals are more noble than other metals. Aluminum 1100, 3003, 3004, 5052, 6053. Web however, you can completely avoid galvanic corrosion by choosing matching metal anchors. Aluminum 1100, 3003,. Web galvanic corrosion (some times called dissimilar metal corrosion) is the process by which the materials in contact with each other oxidizes or corrodes. The position of zinc on the galvanic series, above most other metals, means that it will corrode preferentially if it contacts any of these metals and moisture is present. Web the galvanic series of metals (right). Web what is galvanic corrosion: The closer together the material are on the chart to the right, the less galvanic action will occur. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. The list begins with the more active (anodic) metal and proceeds down. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). Water) is necessary to promote galvanic corrosion. Metals nearer the top of the table are less noble metals and have a greater tendency to lose electrons than the more noble metals found lower on the list. Web because of this, it’s important to understand which metal combinations could create the most risk potential for galvanic corrosion. Aluminum 1100, 3003, 3004, 5052, 6053. Web the galvanic series of metals (right) lists metals and alloys in decreasing order of electrical activity. Web typically, the presence of an electrolyte (eg. Web below is a galvanic reaction chart for dissimilar metals. The chart can be used to determine the likelihood of a galvanic reaction, and galvanic corrosion or bimetallic corrosion, between two different metals in a seawater environment. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. Web however, you can completely avoid galvanic corrosion by choosing matching metal anchors. Water) is necessary to promote galvanic corrosion.
Galvanic Chart Of Metals

Galvanic Corrosion Chart Dissimilar Metals Video Bokep Ngentot
![Galvanic Corrosion [with Chart] EngineerExcel](https://engineerexcel.com/wp-content/uploads/2023/03/galvanic-corrosion-chart.png)
Galvanic Corrosion [with Chart] EngineerExcel
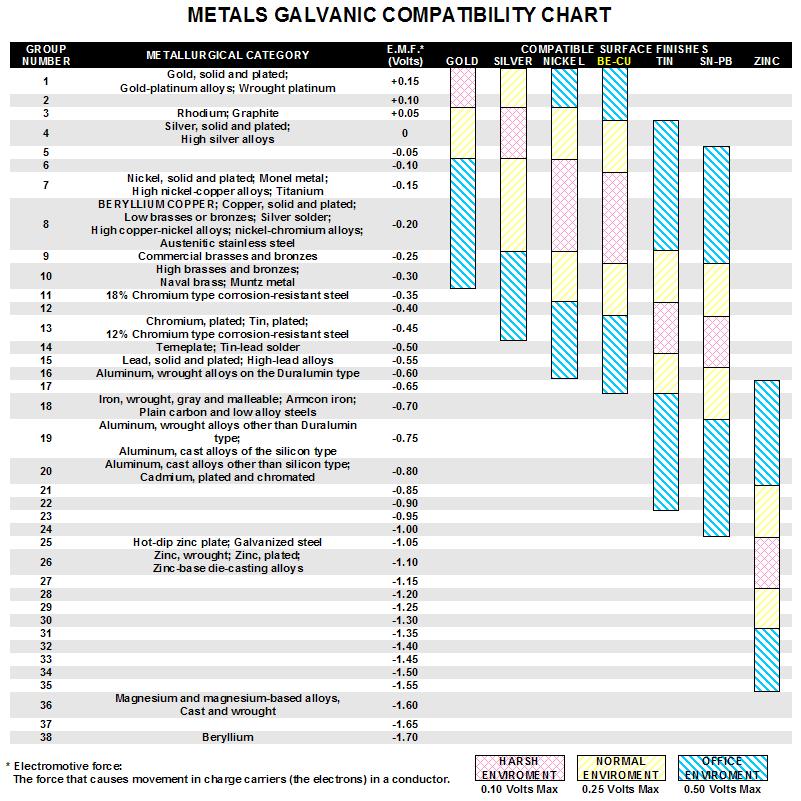
Omega Shielding Products Metals Galvanic Compatibility Chart
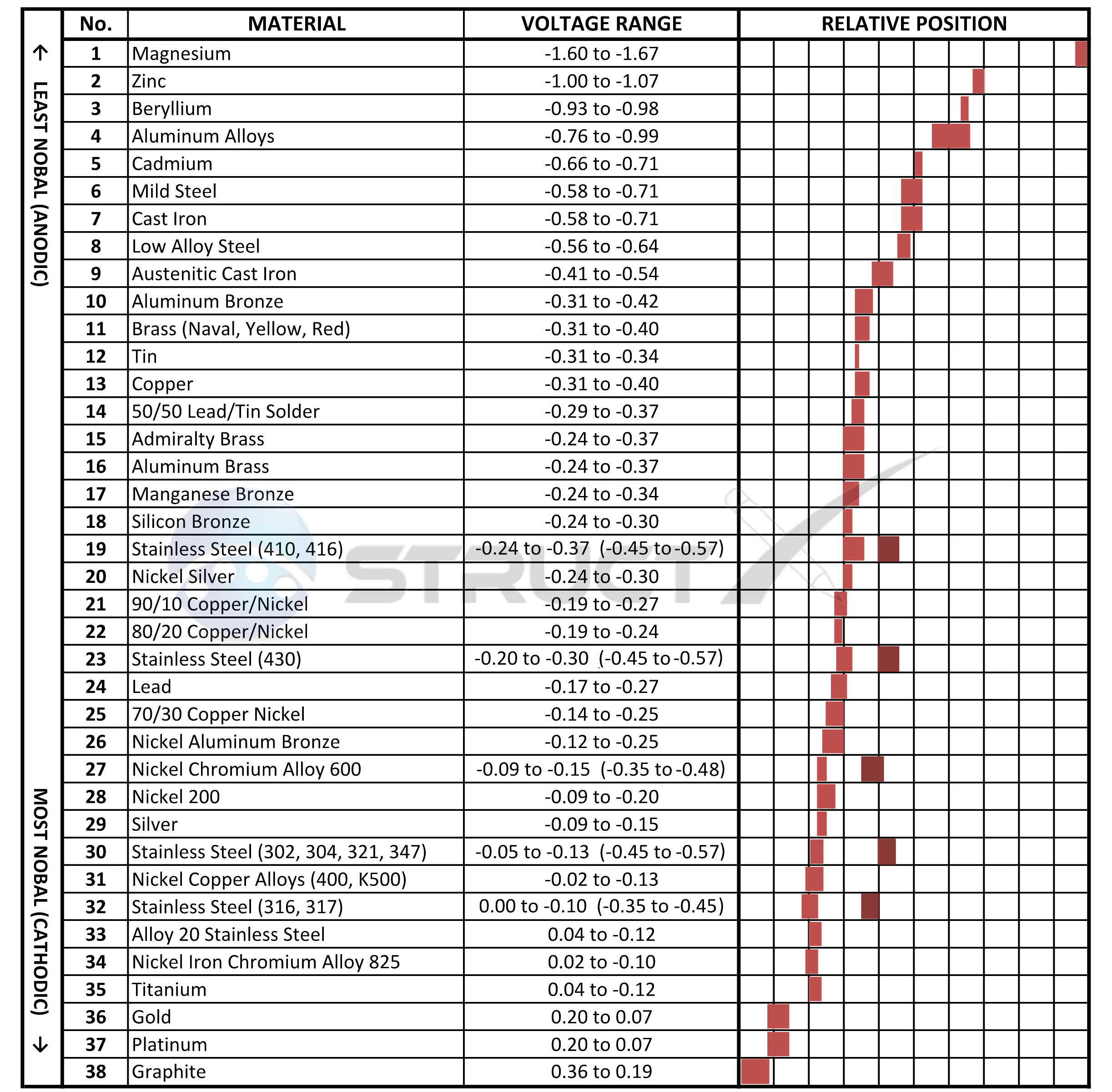
Galvanic Series (electrochemical series)

The Galvanic Series the essential guide EngineeringClicks
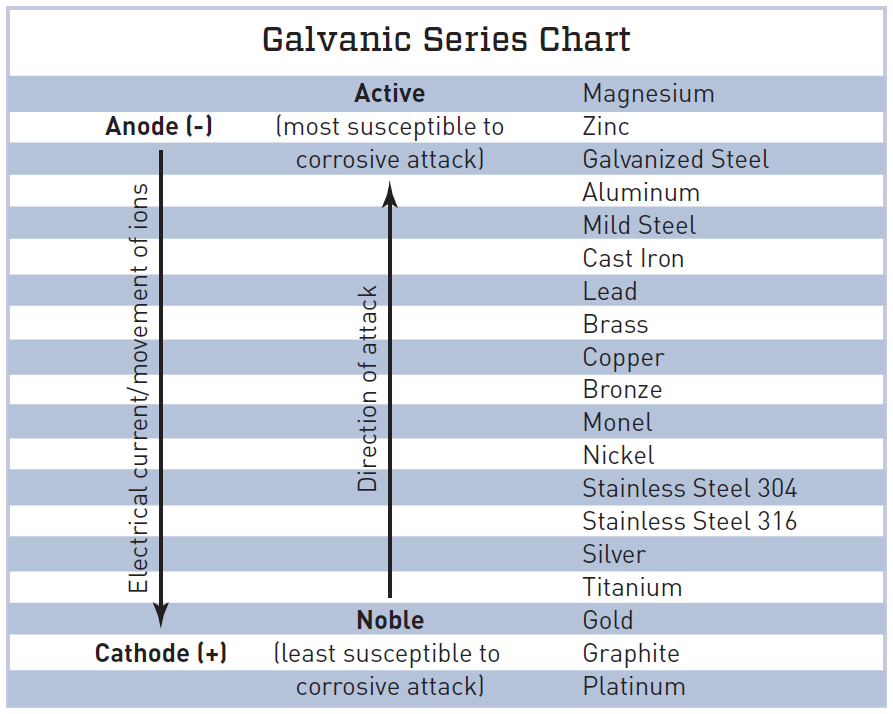
Separating Galvanic Metals JLC Online

An Introduction to the Galvanic Series Galvanic Compatibility and
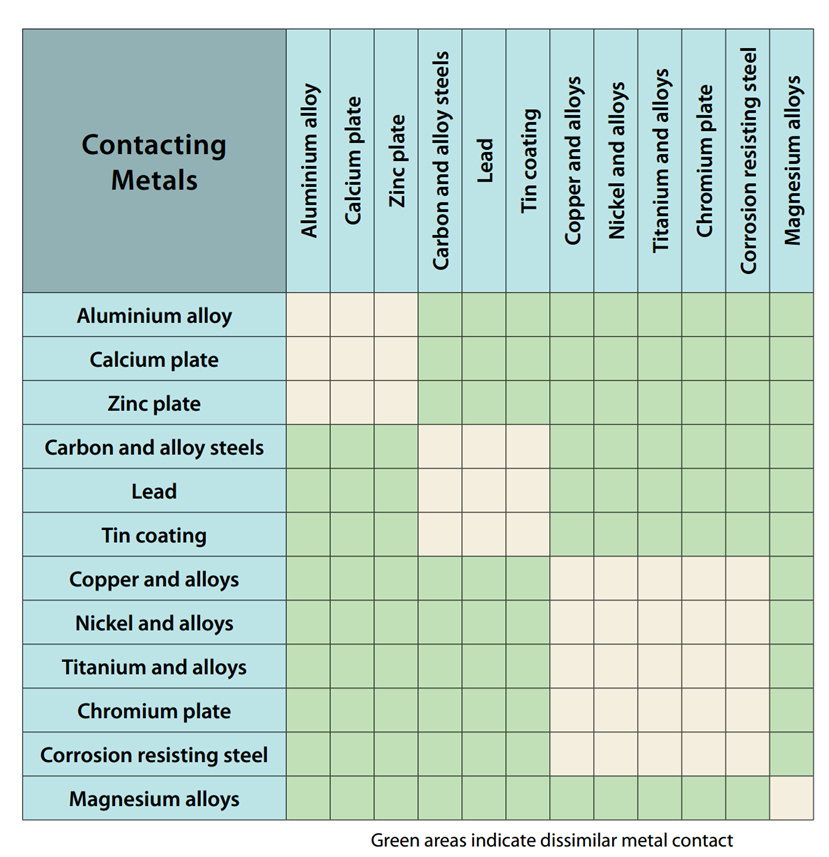
Metals Galvanic Compatibility Chart Online Shopping

Galvanic Chart Of Metals
Web Below Is A Galvanic Reaction Chart For Dissimilar Metals.
Web A Chart Depicting The Galvanic Series For Some Common Metals In A Frequently Encountered Conducting Solution, Seawater, Is Included In Figure 1.
Web Some Metals Like Aluminium And Zinc Develop Tough Oxide Fi Lms.
Galvanic Series Relationships Are Useful As A Guide For Selecting Metals To Be Joined, Will Help The Selection Of Metals Having Minimal Tendency To Interact Galvanically, Or Will Indicate The Need Or Degree Of Protection To Be Applied To Lessen The Expected Potential Interactions.in General, The Further Apart The Materials Are In The Galvanic.
Related Post: