Hybridization And Geometry Chart
Hybridization And Geometry Chart - Determine the hybrid orbitals associated with various molecular geometries Determine the hybridization and bond angles for each. Web here’s a shortcut for how to determine the hybridization of an atom in a molecule that will work in at least 95% of the cases you see in org 1. Find more chemistry widgets in. Want to join the conversation? If it’s 4, your atom is sp3. What orbitals are involved in the tetrahedral arrangement of methane (ch 4 )? Added may 3, 2012 by alessandroserpi in chemistry. Web orbital hybridization involves making linear combinations of the atomic orbitals that are solutions to the schrödinger equation. Sp 2 hybridization results in trigonal geometry. Hybridization of an s orbital with two p orbitals (p x and p y) results in three sp 2 hybrid orbitals that are oriented at 120 o angle to each other (figure 3). Lewis structures provide us with the number and types of bonds around a central atom, as well as any nb electron pairs. Web here’s a shortcut for. Send feedback | visit wolfram|alpha. Why do we need hybridization? Add these two numbers together. Web sp hybridization, bond angle and geometry. Web molecular geometry van koppen/offen. Reactions involve making and breaking of bonds! The arrow points to the vertex of the angle formed. In this video, we use both of these methods to determine the hybridizations of atoms in various organic molecules. Added may 3, 2012 by alessandroserpi in chemistry. Web here’s a shortcut for how to determine the hybridization of an atom in a molecule. Mathematically, this is justified by recognizing that the schrödinger equation is a linear differential equation. Web the five basic shapes of hybridization are linear, trigonal planar, tetrahedral, trigonal bipyramidal and octahedral. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central. Find more chemistry widgets in. Valence electrons repel one another because they are negatively charged and like charges repel. Web summary vsepr and hybridization table. Valence shell electron pair repulsion (vsepr) lewis structures can determine properties such as geometry, bond orders, bond lengths, and dipoles for molecules. Web in sp hybridization, one s orbital and one p orbital hybridize to. So, before we start with organic chemistry, let's revise a few things about bonding in organic molecules. Web in chemistry, orbital hybridisation (or hybridization) is the concept of mixing atomic orbitals to form new hybrid orbitals (with different energies, shapes, etc., than the component atomic orbitals) suitable for the pairing of electrons to form chemical bonds in valence bond theory.. Two electron groups are involved resulting in sp hybridization; In addition to undergrad organic chemistry, this topic is critical for exams like the mcat, gamsat, dat and more. This type of hybridization is required whenever an atom is surrounded by two groups of electrons. Web orbital hybridization involves making linear combinations of the atomic orbitals that are solutions to the. Why do we need hybridization? Valence shell electron pair repulsion (vsepr) lewis structures can determine properties such as geometry, bond orders, bond lengths, and dipoles for molecules. Want to join the conversation? Web orbital hybridization involves making linear combinations of the atomic orbitals that are solutions to the schrödinger equation. Mix at least 2 nonequivalent atomic orbitals (e.g.s and p). Web in sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character. Mix at least 2 nonequivalent atomic orbitals (e.g.s and p). Covalent bonds are formed by: Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to. Web in sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character. This 109.5 o arrangement gives tetrahedral geometry (figure 4). In addition to undergrad organic chemistry, this topic is critical for exams like the mcat, gamsat, dat and more. Web here’s a shortcut for. Covalent bonds are formed by: Web in chemistry, orbital hybridisation (or hybridization) is the concept of mixing atomic orbitals to form new hybrid orbitals (with different energies, shapes, etc., than the component atomic orbitals) suitable for the pairing of electrons to form chemical bonds in valence bond theory. Determine the hybridization and bond angles for each. The arrow points to the vertex of the angle formed. Web energy changes occurring in hybridization. If it’s 4, your atom is sp3. Web the five basic shapes of hybridization are linear, trigonal planar, tetrahedral, trigonal bipyramidal and octahedral. In the last post on the structure of methane we asked how we know that methane is tetrahedral ( see article: Get the free hybridization widget for your website, blog, wordpress, blogger, or igoogle. Web 10.4 hybridization of atomic orbitals 1. Web summary vsepr and hybridization table. Web hybridization increases the overlap of bonding orbitals and explains the molecular geometries of many species whose geometry cannot be explained using a vsepr approach. Lewis structures provide us with the number and types of bonds around a central atom, as well as any nb electron pairs. Hybridization of an s orbital with two p orbitals (p x and p y) results in three sp 2 hybrid orbitals that are oriented at 120 o angle to each other (figure 3). The arrows point to different carbon atoms in the following molecule that are lettered a, b and c. Number of hybrid orbitals is equal to number of pure atomic orbitals used in the hybridization process.
H3O+ Lewis Structure, Geometry, Hybridization, and MO Diagram

Hybridization and Hybrid Orbitals ChemTalk

Hybridization Orbitals Chart
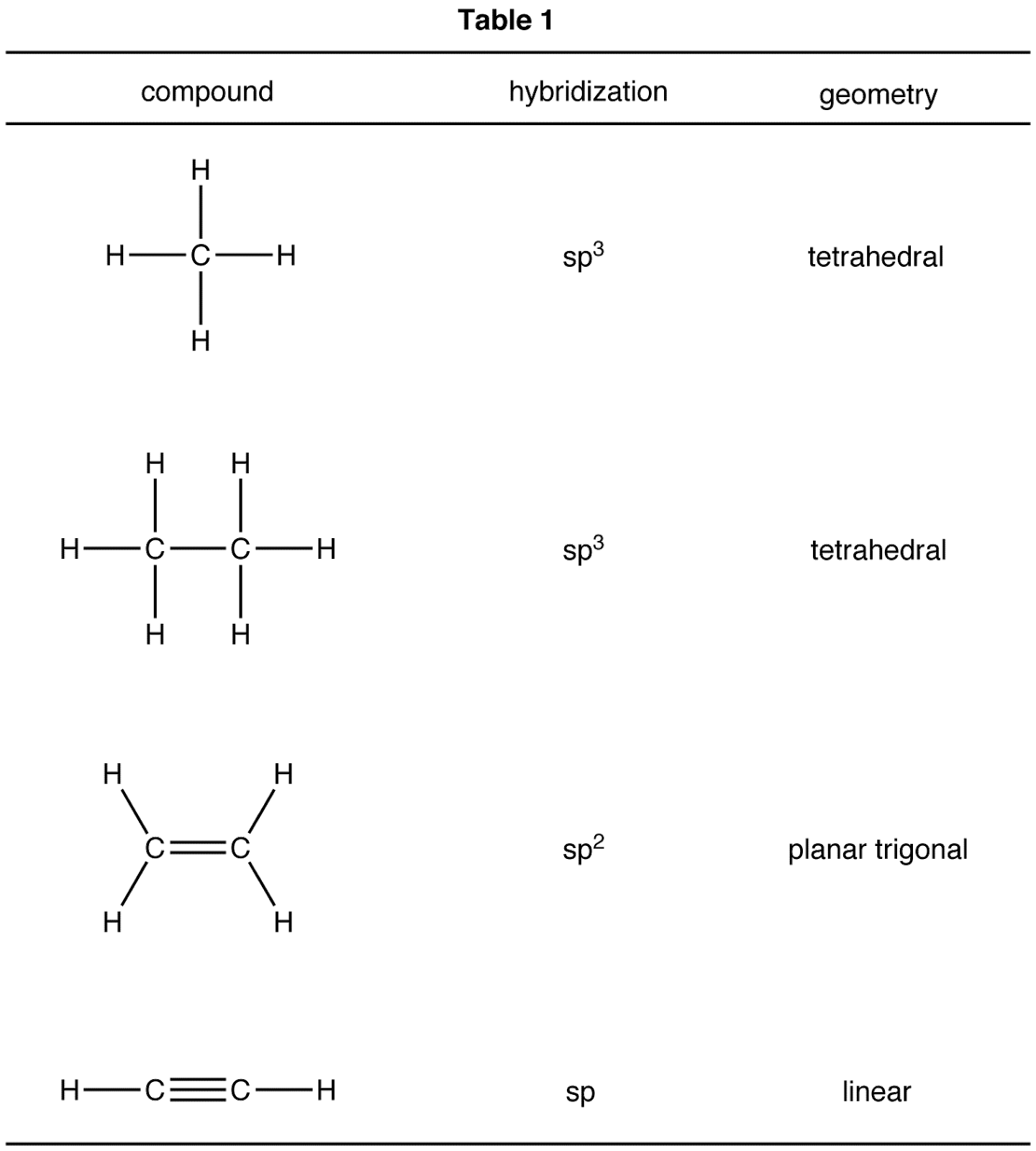
Hybridization And Geometry Chart

C2H4 Lewis Structure, Molecular Geometry, Hybridization, and MO Diagram

MariePreAPChem Hybridization

Molecular Geometry Chart With Hybridization

Molecular Geometry Chart With Hybridization carthopde
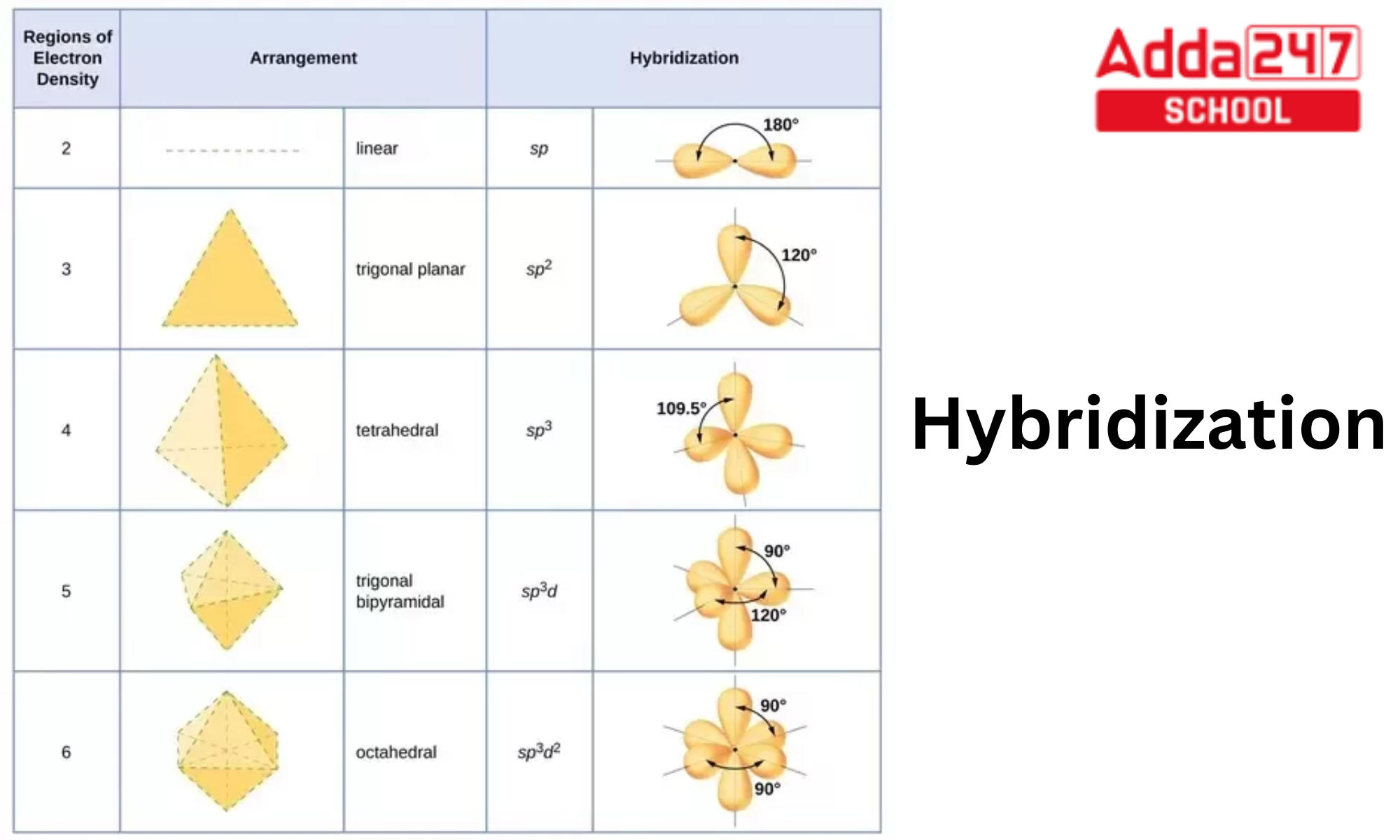
What is Hybridization? sp3, sp2, Examples and Formula
.jpg)
Types of Hybridization Definitions, Examples, Key Features, Steps to
Web Hybridization Of An S Orbital With All Three P Orbitals (P X, P Y, And P Z) Results In Four Sp 3 Hybrid Orbitals.
Valence Electrons Repel One Another Because They Are Negatively Charged And Like Charges Repel.
Send Feedback | Visit Wolfram|Alpha.
Reactions Involve Making And Breaking Of Bonds!
Related Post: