Hybridization Geometry Chart
Hybridization Geometry Chart - The geometry of the orbital arrangement is as follows: Web 10.4 hybridization of atomic orbitals 1. How does molecule shape change with different numbers of bonds and electron pairs? Find more chemistry widgets in. Sp 2 hybridization results in trigonal geometry. Web here’s a shortcut for how to determine the hybridization of an atom in a molecule that will work in at least 95% of the cases you see in org 1. Then, compare the model to real molecules! Web the hybridization of an atom is determined based on the number of regions of electron density that surround it. Web the five basic shapes of hybridization are linear, trigonal planar, tetrahedral, trigonal bipyramidal and octahedral. Number of hybrid orbitals is equal to number of pure atomic orbitals used in the hybridization process. Web sp³, sp² and sp hybridization, or the mixing of s and p orbitals which allows us to create sigma and pi bonds, is a topic we usually think we understand, only to get confused when it reappears in organic chemistry molecules and reactions. Get the free hybridization widget for your website, blog, wordpress, blogger, or igoogle. Here is a. Find out by adding single, double or triple bonds and lone pairs to the central atom. Ways carbon can bond to others. Mix at least 2 nonequivalent atomic orbitals (e.g.s and p). Web here’s a shortcut for how to determine the hybridization of an atom in a molecule that will work in at least 95% of the cases you see. Web the five basic shapes of hybridization are linear, trigonal planar, tetrahedral, trigonal bipyramidal and octahedral. This 109.5 o arrangement gives tetrahedral geometry (figure 4). Web sp³, sp² and sp hybridization, or the mixing of s and p orbitals which allows us to create sigma and pi bonds, is a topic we usually think we understand, only to get confused. The geometry of the orbital arrangement is as follows: Get the free hybridization widget for your website, blog, wordpress, blogger, or igoogle. Find out by adding single, double or triple bonds and lone pairs to the central atom. Web we can find the hybridization of an atom in a molecule by either looking at the types of bonds surrounding the. Send feedback | visit wolfram|alpha. Web we can find the hybridization of an atom in a molecule by either looking at the types of bonds surrounding the atom or by calculating its steric number. Valence electrons repel one another because they are negatively charged and like charges repel. Predicted bond angle(s) hybridization of central atom. This type of hybridization is. Web hybridization is the idea that atomic orbitals fuse to form newly hybridized orbitals, which in turn, influences molecular geometry and bonding properties. In this video, we use both of these methods to determine the hybridizations of atoms in various organic molecules. Web hybridization increases the overlap of bonding orbitals and explains the molecular geometries of many species whose geometry. Added may 3, 2012 by alessandroserpi in chemistry. An atom has a given hybridization depending on the number of bonds extending from it. Here is a chart that sums this up: Web hybridization increases the overlap of bonding orbitals and explains the molecular geometries of many species whose geometry cannot be explained using a vsepr approach. There is also an. An atom has a given hybridization depending on the number of bonds extending from it. Web learn the definition of orbital hybridization and the characteristics and geometries of sp, sp2, sp3, sp3d1, and sp3d2 hybridization. This type of hybridization is required whenever an atom is surrounded by two groups of electrons. Reactions involve making and breaking of bonds! Web the. How does molecule shape change with different numbers of bonds and electron pairs? Web in sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character. Explore molecule shapes by building molecules in 3d! Web hybridization of an s orbital with two p orbitals (p x. Ways carbon can bond to others. Web hybridization increases the overlap of bonding orbitals and explains the molecular geometries of many species whose geometry cannot be explained using a vsepr approach. Web molecular geometry van koppen/offen procedure: If it’s 4, your atom is sp3. There is also an implicit geometric shape associated with the hybridization. Web 10.4 hybridization of atomic orbitals 1. Valence electrons repel one another because they are negatively charged and like charges repel. The angle between the orbitals is 180°. Web here’s a shortcut for how to determine the hybridization of an atom in a molecule that will work in at least 95% of the cases you see in org 1. There is also an implicit geometric shape associated with the hybridization. The geometry of the orbital arrangement is as follows: Sp 2 hybridization results in trigonal geometry. The geometrical arrangements characteristic of the various sets of hybrid orbitals are shown in figure 8.21. Explore molecule shapes by building molecules in 3d! This type of hybridization is required whenever an atom is surrounded by two groups of electrons. Web sp³, sp² and sp hybridization, or the mixing of s and p orbitals which allows us to create sigma and pi bonds, is a topic we usually think we understand, only to get confused when it reappears in organic chemistry molecules and reactions. Added may 3, 2012 by alessandroserpi in chemistry. Get the free hybridization widget for your website, blog, wordpress, blogger, or igoogle. This 109.5 o arrangement gives tetrahedral geometry (figure 4). Hybrid orbitals have very different shape from original atomic orbitals. Web in sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character.
XeCl4 Lewis Structure, Geometry, Hybridization, and Polarity

Molecular Geometry Chart With Hybridization carthopde

Hybridization Wize University Chemistry Textbook Wizeprep
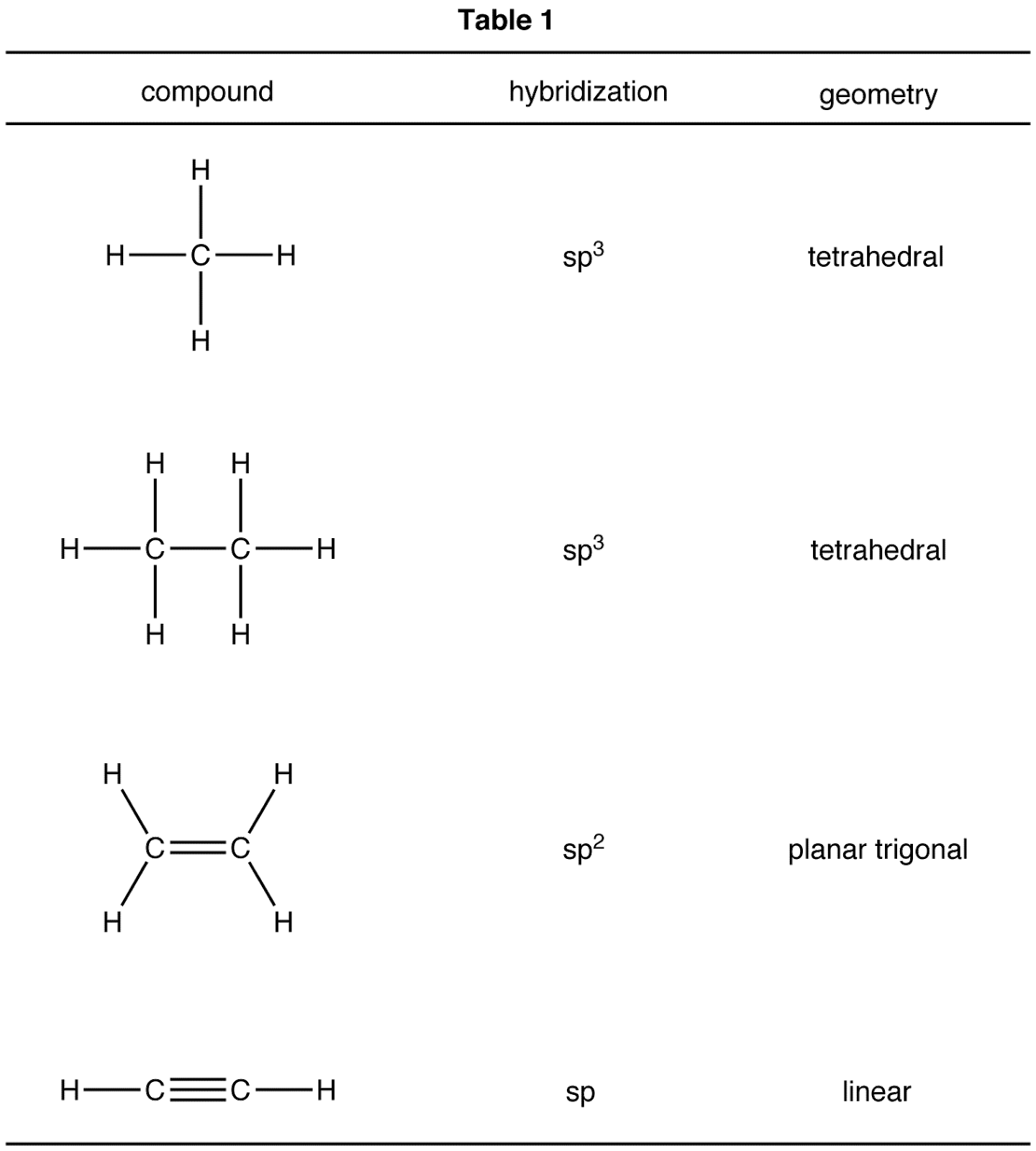
Molecular Geometry And Hybridization Chart

MariePreAPChem Hybridization
.jpg)
Types of Hybridization Definitions, Examples, Key Features, Steps to
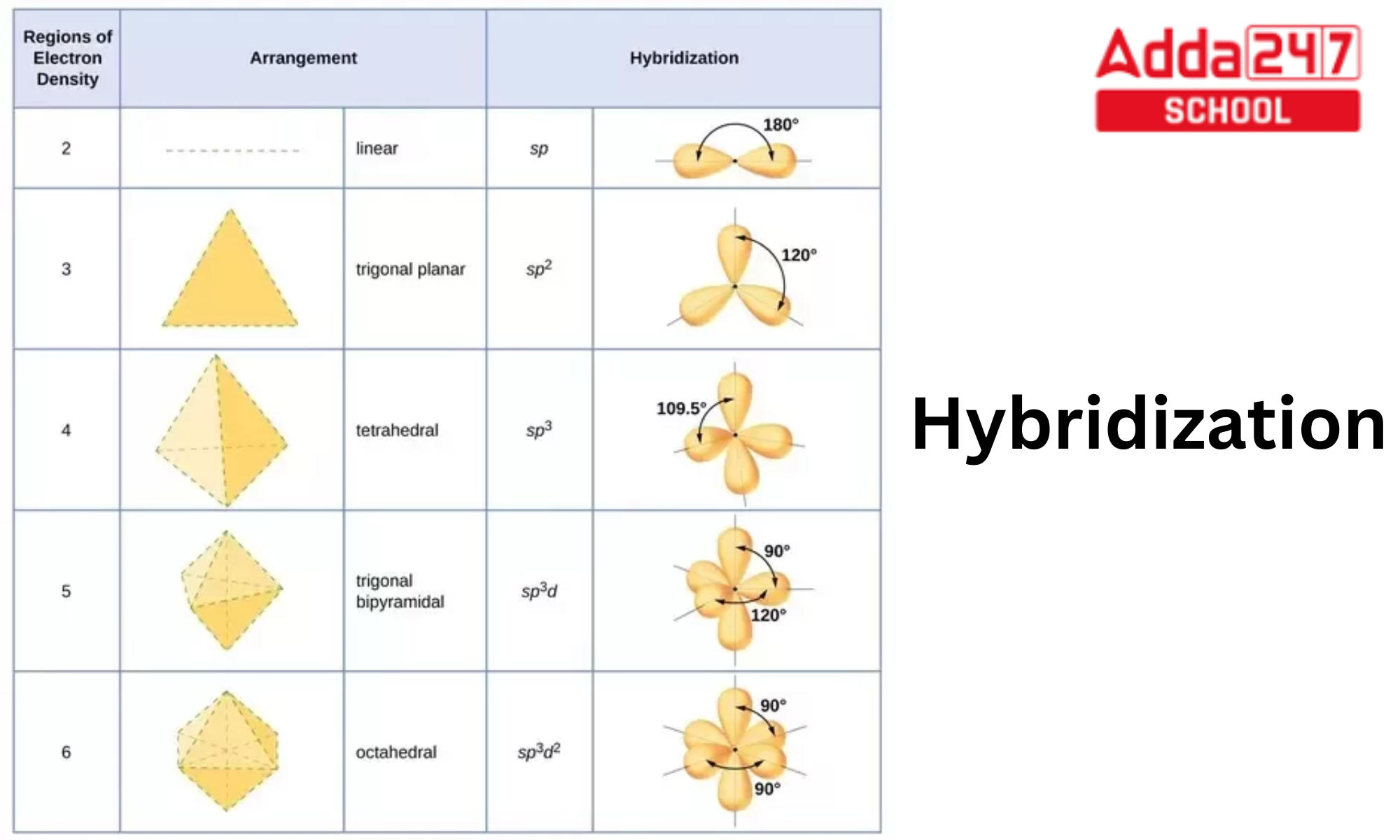
What is Hybridization? sp3, sp2, Examples and Formula

Hybridization and Hybrid Orbitals ChemTalk

Hybridization Orbitals Chart

Molecular Geometry Chart With Hybridization
Here Is A Chart That Sums This Up:
Web The Five Basic Shapes Of Hybridization Are Linear, Trigonal Planar, Tetrahedral, Trigonal Bipyramidal And Octahedral.
Send Feedback | Visit Wolfram|Alpha.
Web We Recommend Using The Latest Version Of Chrome, Firefox, Safari, Or Edge.
Related Post: