Nitrogen Gas Temperature Pressure Chart
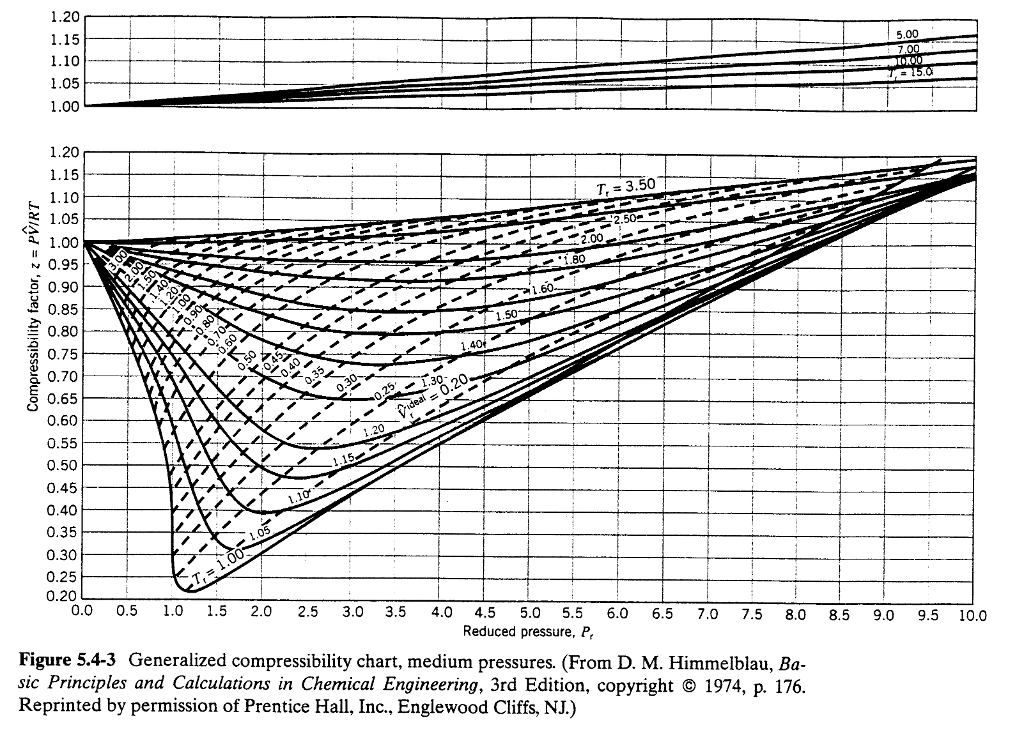
Nitrogen Gas Temperature Pressure Chart - Ideal gas law equation ideal gas constant faqs. What is an ideal gas? Web thermophysical properties of fluid systems. This ideal gas law calculator will help you establish the properties of an ideal gas subject to pressure, temperature, or volume changes. Dalton's law is the final law, and it states that the combined pressure of all gases in a closed space is. The calculation assumes that the amount and volume of gas do not change. Web temperature is sometimes measured with a gas thermometer by observing the change in the volume of the gas as the temperature changes at constant pressure. Please choose the units you wish to use: The gas constant is r = 8314.3 j/ k'kmol. Streng, 1971 streng, a.g., miscibility and compatibility of some liquid and solidified gases at low temperature, j. This structure is also available as a 2d mol file or as a computed 3d sd file. Streng, 1971 streng, a.g., miscibility and compatibility of some liquid and solidified gases at low temperature, j. What is an ideal gas? Web the equations describing these laws are special cases of the ideal gas law, pv = nrt, where p is the. Please choose the units you wish to use: The molecular weight is m = 28.016 kg/kmol. Streng, 1971 streng, a.g., miscibility and compatibility of some liquid and solidified gases at low temperature, j. This structure is also available as a 2d mol file or as a computed 3d sd file. Streng, 1971 streng, a.g., miscibility and compatibility of some liquid. The hydrogen in a particular hydrogen gas thermometer has a volume of 150.0 cm 3 when immersed in a mixture of ice and water (0.00 °c). Jahangiri, m., termodynamic properties of nitrogen from the freezing line to 2000 k at pressures to 1000 mpa, j. Web it should be noted that n« gas can be considered as an ideal gas. Please follow the steps below to select the data required. Streng, 1971 streng, a.g., miscibility and compatibility of some liquid and solidified gases at low temperature, j. These data include the following: Ideal gas law equation ideal gas constant faqs. Web temperature is sometimes measured with a gas thermometer by observing the change in the volume of the gas as. Web the compressibility factor of an ideal gas is exactly one. Calculation of thermodynamic state variables of nitrogen in saturation state, boiling curve. Accurate thermophysical properties are available for several fluids. Web lower limit for calculation: What is an ideal gas? This structure is also available as a 2d mol file or as a computed 3d sd file. Accurate thermophysical properties are available for several fluids. Please be patient while the web interface loads. We then have the law for an ideal gas: This ideal gas law calculator will help you establish the properties of an ideal gas subject to pressure,. Jahangiri, m., termodynamic properties of nitrogen from the freezing line to 2000 k at pressures to 1000 mpa, j. Web compute the values of pressure of a gas for various temperatures using the entered temperature and the known value of pressure at that temperature. Web table of contents. Please follow the steps below to select the data required. Streng, 1971. Calculation of thermodynamic state variables of nitrogen in saturation state, boiling curve. These data include the following: Please select the species of interest : Streng, 1971 streng, a.g., miscibility and compatibility of some liquid and solidified gases at low temperature, j. Thermal diffusivity of nitrogen at 1, 10, 50 and 100 bara (14.5, 145, 725 and 1450 psia), and varying. What is an ideal gas? The gas constant is r = 8314.3 j/ k'kmol. Figures 3.2.1 and 3.2.2 illustrate the compressibility factors of hydrogen and nitrogen, respectively, over a. Thermal diffusivity of nitrogen at 1, 10, 50 and 100 bara (14.5, 145, 725 and 1450 psia), and varying temperature given as °c or °f: Web the compressibility factor of an. Web thermophysical properties of fluid systems. The gas constant is r = 8314.3 j/ k'kmol. Web temperature is sometimes measured with a gas thermometer by observing the change in the volume of the gas as the temperature changes at constant pressure. Calculation of thermodynamic state variables of nitrogen in saturation state, boiling curve. Dalton's law is the final law, and. Please select the species of interest : In this way table 5, rg versus p and t, can be extended beyond t = 350k. These data include the following: Web it should be noted that n« gas can be considered as an ideal gas for temperatures t > 350k and pressures p <, 10 bar. Web temperature is sometimes measured with a gas thermometer by observing the change in the volume of the gas as the temperature changes at constant pressure. Calculation of thermodynamic state variables of nitrogen in saturation state, boiling curve. Jahangiri, m., termodynamic properties of nitrogen from the freezing line to 2000 k at pressures to 1000 mpa, j. Ideal gas law equation ideal gas constant faqs. For real gases, the compressibility factor may be very different from one. Web the equations describing these laws are special cases of the ideal gas law, pv = nrt, where p is the pressure of the gas, v is its volume, n is the number of moles of the gas, t is its kelvin temperature, and r is the ideal (universal) gas constant. Web table of contents. The gas constant is r = 8314.3 j/ k'kmol. Web early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and temperature, for example), varying a third (such as pressure), and measuring the effect of the change on the fourth (in this case, volume). Jahangiri, m., termodynamic properties of nitrogen from the freezing line to 2000 k at pressures to 1000 mpa, j. Web thermophysical properties of fluid systems. Please choose the units you wish to use:
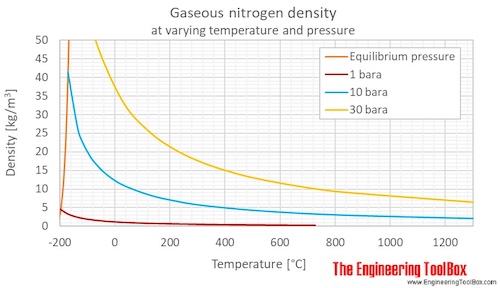
Density of nitrogen with change in temperature and at different

Nitrogen Thermal Diffusivity vs. Temperature and Pressure

Nitrogen Thermal Conductivity vs. Temperature and Pressure

Nitrogen Pressure Temperature Chart A Visual Reference of Charts
Nitrogen Pressure Chart A Visual Reference of Charts Chart Master

Nitrogen Enthalpy, Internal Energy and Entropy vs. Temperature

Nitrogen Thermal Diffusivity vs. Temperature and Pressure

Nitrogen Density and Specific Weight vs. Temperature and Pressure

Nitrogen Pressure Temperature Chart
Nitrogen Pressure Vs Temperature Chart
This Structure Is Also Available As A 2D Mol File Or As A Computed 3D Sd File.
The 3D Structure May Be Viewed Using Java Or Javascript.
The Hydrogen In A Particular Hydrogen Gas Thermometer Has A Volume Of 150.0 Cm 3 When Immersed In A Mixture Of Ice And Water (0.00 °C).
The Calculation Assumes That The Amount And Volume Of Gas Do Not Change.
Related Post: