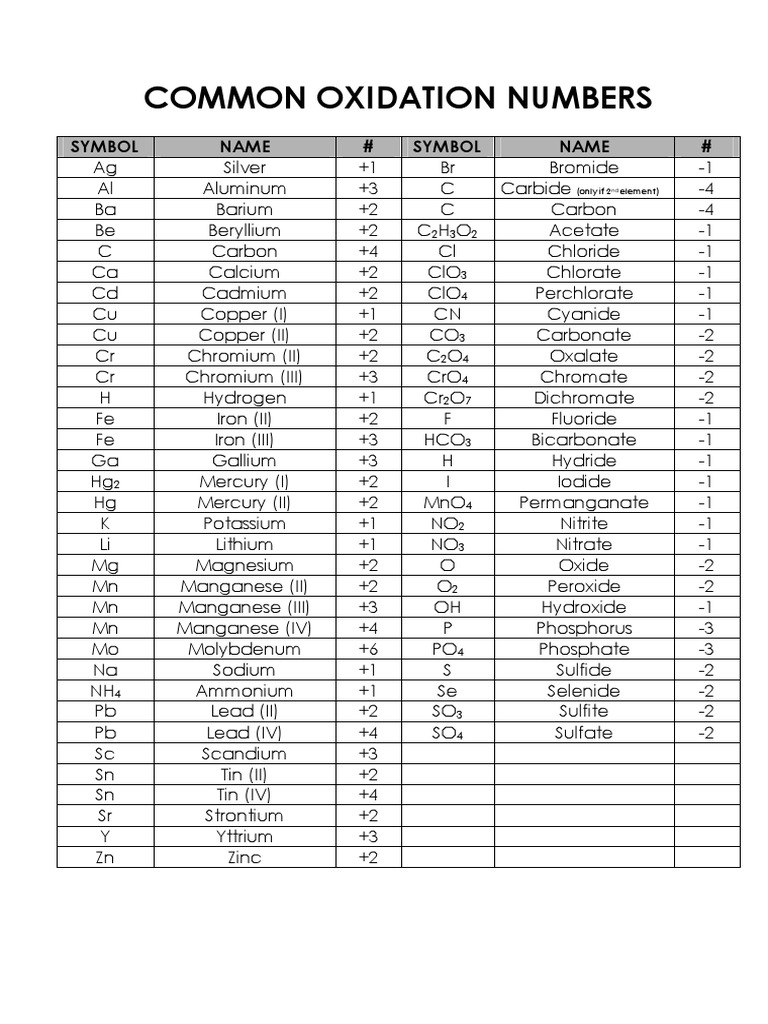
Oxidation Numbers Chart
Oxidation Numbers Chart - Name cart, relative atomic mass chart, electronegativity chart and much more. Web list of oxidation states of the elements. Web it’s essentially a number assigned to an element in a compound, and (at a basic level) denotes the number of electrons that have been either removed from or. Web enter the formula of a chemical compound to find the oxidation number of each element. Web determine what is the oxidizing and reducing agents in the following reaction. More than one oxidation numbers of an element. Web an oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction. This is a list of all the known oxidation states of the chemical elements, excluding nonintegral values. The oxidation number of an element in its free state is zero. Zn + 2h+ zn2+ +h2 zn + 2 h + zn 2 + + h 2. Web the oxidation state of an atom is equal to the total number of electrons which have been removed from an element (producing a positive oxidation state) or added to an element. More than one oxidation numbers of an element. Web oxidation state shows the total number of electrons which have been removed from an element (a positive oxidation state). Lih, nah, and cah 2. Web learn what oxidation numbers are, how they relate to valence electrons, bonding, and electron configuration. Web enter the formula of a chemical compound to find the oxidation number of each element. Redox reactions are characterized by a. See examples, rules, and exceptions for different elements and. Web enter the formula of a chemical compound to find the oxidation number of each element. Web list of oxidation states of the elements. Lih, nah, and cah 2. How to download chemix school. Zn + 2h+ zn2+ +h2 zn + 2 h + zn 2 + + h 2. We can use oxidation numbers to keep track of where electrons are in a molecule, and how. The oxidation number of an element in its free state is zero. Web the oxidation state of an atom is equal to the total number of electrons which have been removed from an element (producing a positive oxidation state) or added to an. The term oxidation state is often used. Name cart, relative atomic mass chart, electronegativity chart and much more. Web it’s essentially a number assigned to an element in a compound, and (at a basic level) denotes the number of electrons that have been either removed from or. We can use oxidation numbers to keep track of where electrons are in. Web the oxidation number is the positive or negative number of an atom that indicates the electrical charge the atom has if its compound consists of ions. Web oxidation state shows the total number of electrons which have been removed from an element (a positive oxidation state) or added to an element (a negative oxidation state). See examples, rules, and. Web the oxidation state of an atom is equal to the total number of electrons which have been removed from an element (producing a positive oxidation state) or added to an element. The oxidation number of a monatomic. Web an oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation. See the name, symbol and oxidation number of each element in a table format. More than one oxidation numbers of an element. Zn + 2h+ zn2+ +h2 zn + 2 h + zn 2 + + h 2. Mno2 wouldn't be considered an oxyanion. The term oxidation state is often used. Web determine what is the oxidizing and reducing agents in the following reaction. More than one oxidation numbers of an element. Zn + 2h+ zn2+ +h2 zn + 2 h + zn 2 + + h 2. This is a list of all the known oxidation states of the chemical elements, excluding nonintegral values. Web the oxidation state of an. More than one oxidation numbers of an element. Web oxidation state shows the total number of electrons which have been removed from an element (a positive oxidation state) or added to an element (a negative oxidation state). Ch 4, nh 3, h 2 o, and hcl. Web data chart of the oxidation states of the chemical elements. Web 60 rows. Ch 4, nh 3, h 2 o, and hcl. Web the oxidation state of an atom is equal to the total number of electrons which have been removed from an element (producing a positive oxidation state) or added to an element. The oxidation number of an element in its free state is zero. Name cart, relative atomic mass chart, electronegativity chart and much more. Web data chart of the oxidation states of the chemical elements. Web determine what is the oxidizing and reducing agents in the following reaction. Oxidation number of an atom can be positive or negative or may be zero. Web 60 rows find the oxidation numbers of monoatomic ions for each element in the periodic table. Web oxidation state shows the total number of electrons which have been removed from an element (a positive oxidation state) or added to an element (a negative oxidation state). More than one oxidation numbers of an element. Mno2 wouldn't be considered an oxyanion. Web list of oxidation states of the elements. The term oxidation state is often used. Web learn what oxidation numbers are, how they relate to valence electrons, bonding, and electron configuration. Zn + 2h+ zn2+ +h2 zn + 2 h + zn 2 + + h 2. Web as you see from the picture above you can also view :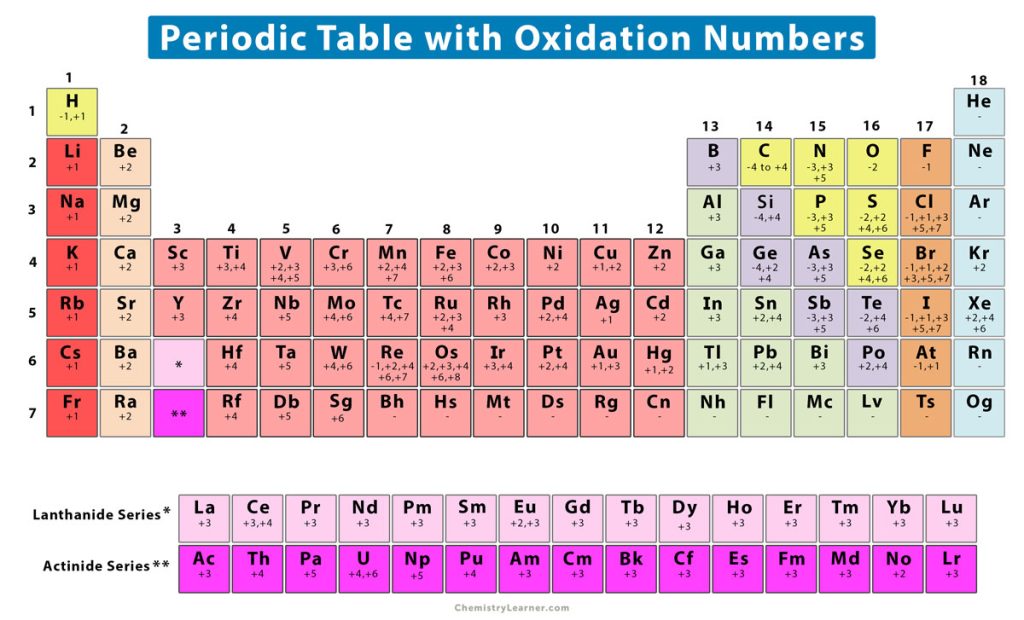
Oxidation Number (State) Definition, Rules, How to Find, and Examples

Periodic Table Oxidation Chart
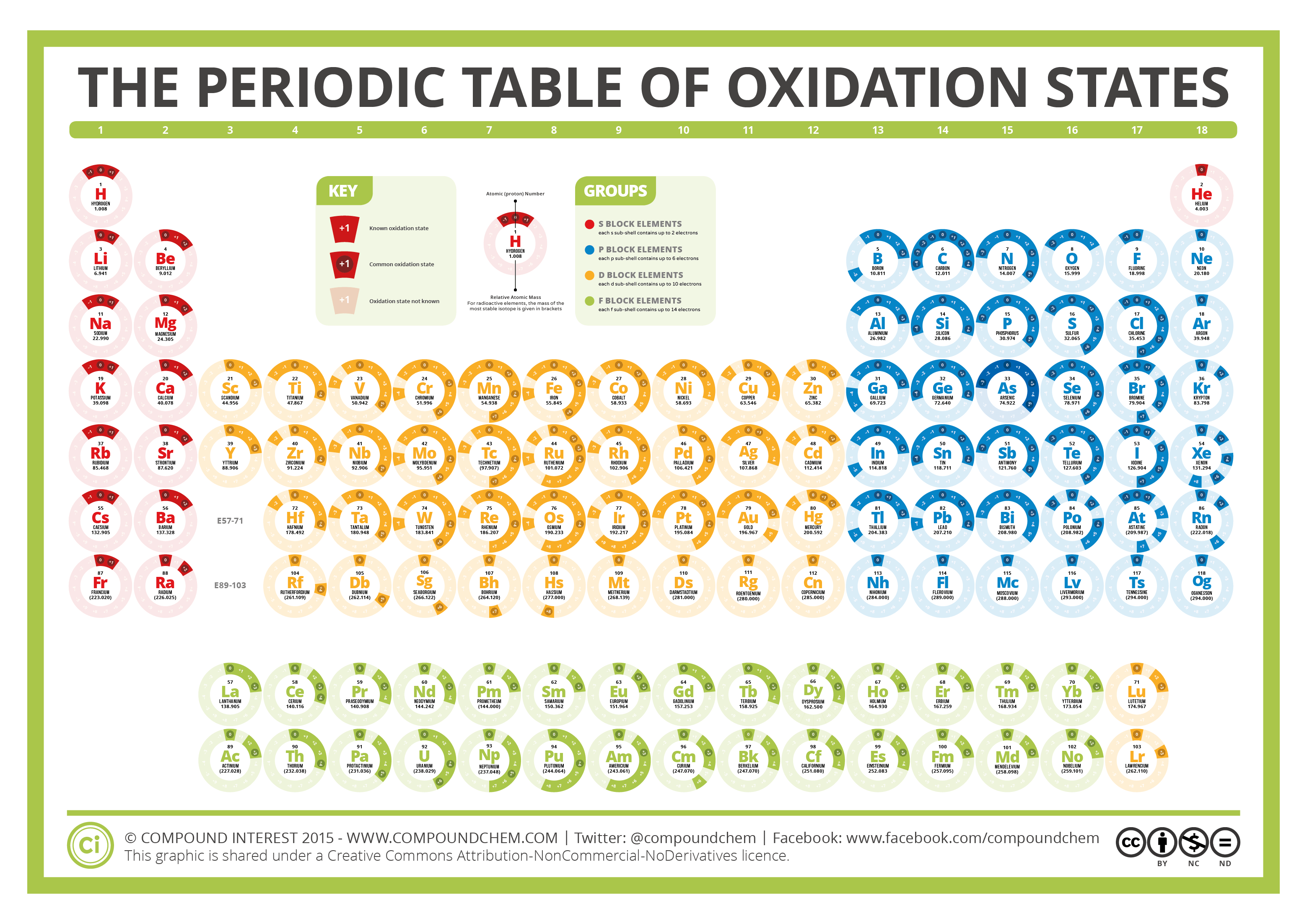
The Periodic Table of Oxidation States Compound Interest
Periodic Table Of Elements With Oxidation Numbers Hot Sex Picture
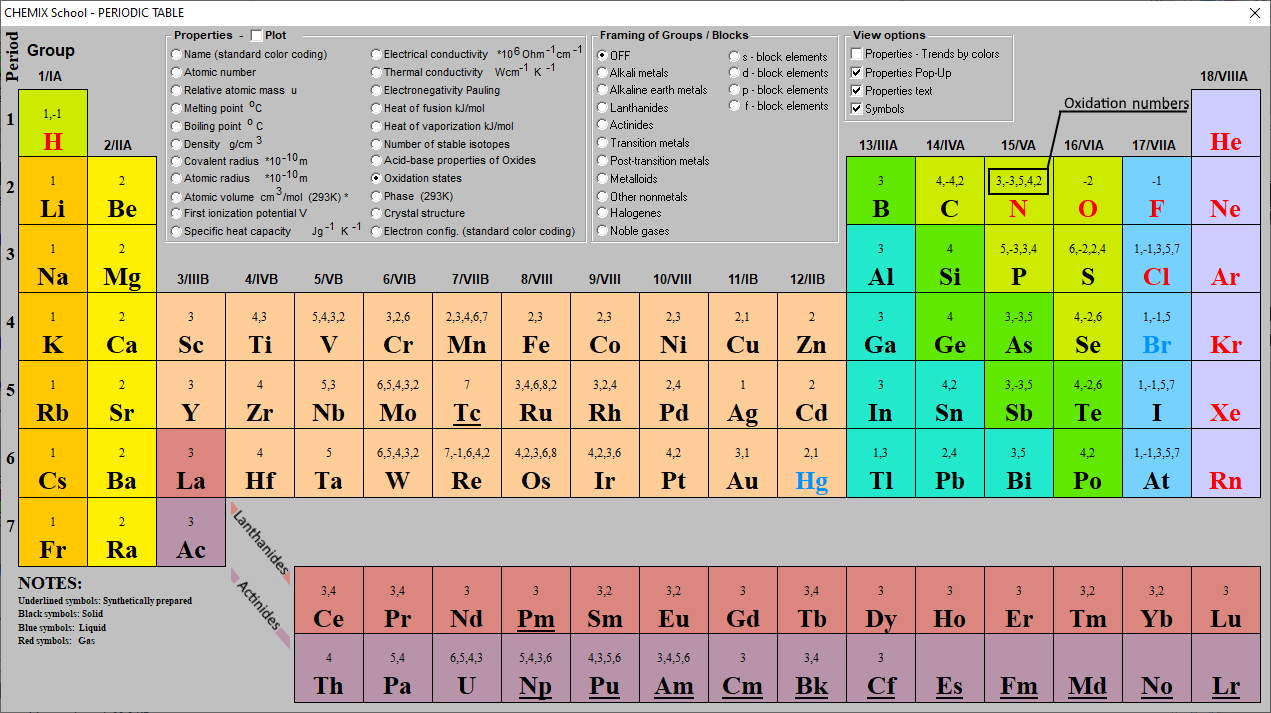
Oxidation Numbers Periodic Table Elements
Common Oxidation Numbers Chart
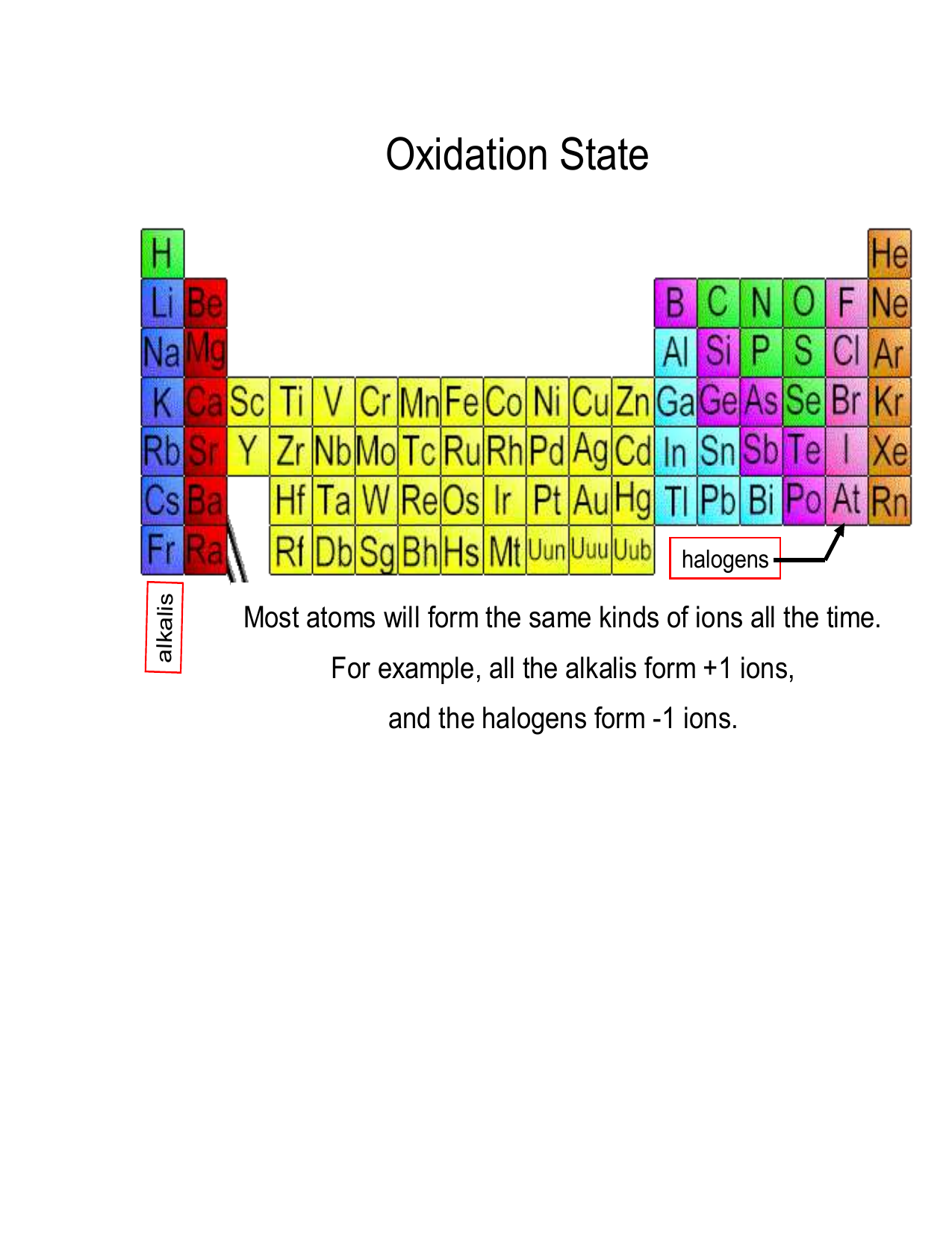
Oxidation Numbers

Oxidation
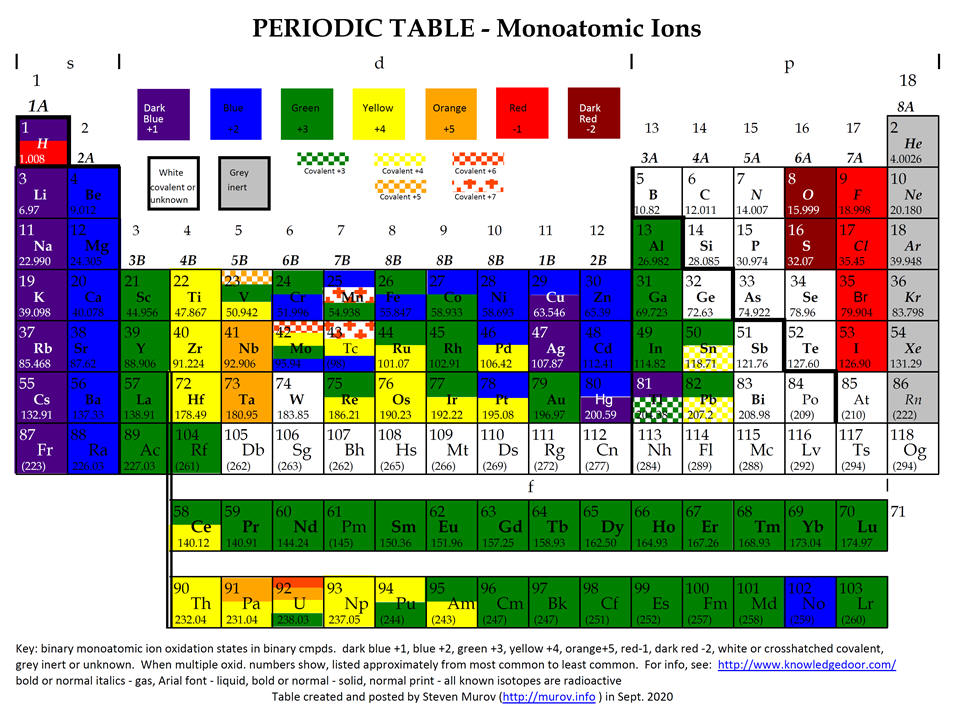
Chemistry Insight

Downloadable Periodic Table Oxidation States
The Oxidation Number Of A Monatomic.
Important Facts Of Oxidation Numbers.
Web It’s Essentially A Number Assigned To An Element In A Compound, And (At A Basic Level) Denotes The Number Of Electrons That Have Been Either Removed From Or.
Lih, Nah, And Cah 2.
Related Post: