Ph To Poh Chart
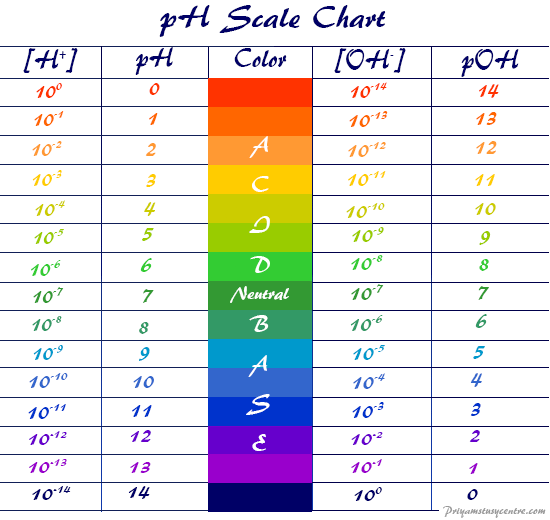
Ph To Poh Chart - The danish biochemist søren sørenson proposed the term ph. Web with this ph calculator, you can determine the ph of a solution in a few ways. Hydrogen & hydroxide ion concentration calculator. Ph = 14 − poh = 14 − 10.5 = 3.5. The first column is labeled “left bracket h subscript 3 o superscript plus right bracket (m).” This is because of rounding off. Express hydronium and hydroxide ion concentrations on the ph and poh scales. You can use ph p h to make a quick determination whether a. Poh is related to ph and can be easily calculated from ph. The ph scale is the range of value s from 0 to 14 that describes the acidity or basicity of a solution. By the end of this section, you will be able to: For a solution with a poh of 10.5, the corresponding ph is approximately 3.5. To calculate ph, first convert concentration to molarity. The ph value is an essential factor in chemistry, medicine, and daily life. Read the text below to find out what is the ph scale and the. By the end of this section, you will be able to: Web when a ph, a poh, or k w is shown, no unit of m will appear. Web if ph > 7, then the solution is basic. Hydrogen & hydroxide ion concentration calculator. Here is a table of ph values of common chemicals. Web in a chemistry problem, you may be given concentration in other units. Perform calculations relating ph and poh. Perform calculations relating ph and poh. Explain the characterization of aqueous solutions as acidic, basic, or neutral. The ph value is an essential factor in chemistry, medicine, and daily life. As we have seen, [h3o+] [ h 3 o +] and [oh−] [ o h −] values can be markedly different from one aqueous solution to another. Express hydronium and hydroxide ion concentrations on the ph and poh scales. Determine the ph of acidic and basic solutions. By the end of this section, you will be able to: Explain the. Exponential numbers should be entered using e to represent 10^. (they are both h + ion, or proton, donors.) but they differ in the extent to which they donate h + ions to water. Perform calculations relating ph and poh. These examples demonstrate the straightforward conversion between ph and poh using the relationship ph + poh = 14 in aqueous. Hydrogen & hydroxide ion concentration calculator. Web in a chemistry problem, you may be given concentration in other units. Web if ph > 7, then the solution is basic. The poh calculator simplifies this process by allowing you to quickly determine the poh value based on the hydroxide ion concentration. Perform calculations relating ph and poh. Express hydronium and hydroxide ion concentrations on the ph and poh scales. It can convert ph to h +, as well as calculate ph from the ionization constant and concentration. Exponential numbers should be entered using e to represent 10^. Web with this ph calculator, you can determine the ph of a solution in a few ways. These examples demonstrate. Determine the ph of acidic and basic solutions. Determine the hydronium ion concentration and poh from ph. Perform calculations relating ph and poh. Ph = 14 − poh = 14 − 10.5 = 3.5. Web with this ph calculator, you can determine the ph of a solution in a few ways. This is because of rounding off. By the end of this section, you will be able to: The ph scale is the range of value s from 0 to 14 that describes the acidity or basicity of a solution. Web both compounds satisfy the brnsted definition of an acid. Determine the hydronium ion concentration and poh from ph. Web ph and poh are defined as the negative log of hydrogen ion concentration and hydroxide concentration, respectively. Express hydronium and hydroxide ion concentrations on the ph and poh scales. To calculate ph, first convert concentration to molarity. This is because of rounding off. Calculating the ph of a strong acid or base solution. Determine the hydronium ion concentration and poh from ph. This is because of rounding off. Chemicals with ph values from 0 up to 7 are acids, those with a ph value of 7 are neutral, and those with ph values greater than 7 up to 14 are bases. Calculating the poh (power of hydroxide ion concentration) is essential in chemistry, especially when dealing with solutions and their basicity. Explain the characterization of aqueous solutions as acidic, basic, or neutral. Calculating the ph of a strong acid or base solution. (they are both h + ion, or proton, donors.) but they differ in the extent to which they donate h + ions to water. By definition, a strong acid is any substance that is good at donating an h + ion to water. By the end of this section, you will be able to: Web if ph > 7, then the solution is basic. Web it indicates how acidic or basic a solution is. By the end of this module, you will be able to: Web define ph and poh. Here is a table of ph values of common chemicals. Express hydronium and hydroxide ion concentrations on the ph and poh scales. The ph scale is the range of value s from 0 to 14 that describes the acidity or basicity of a solution.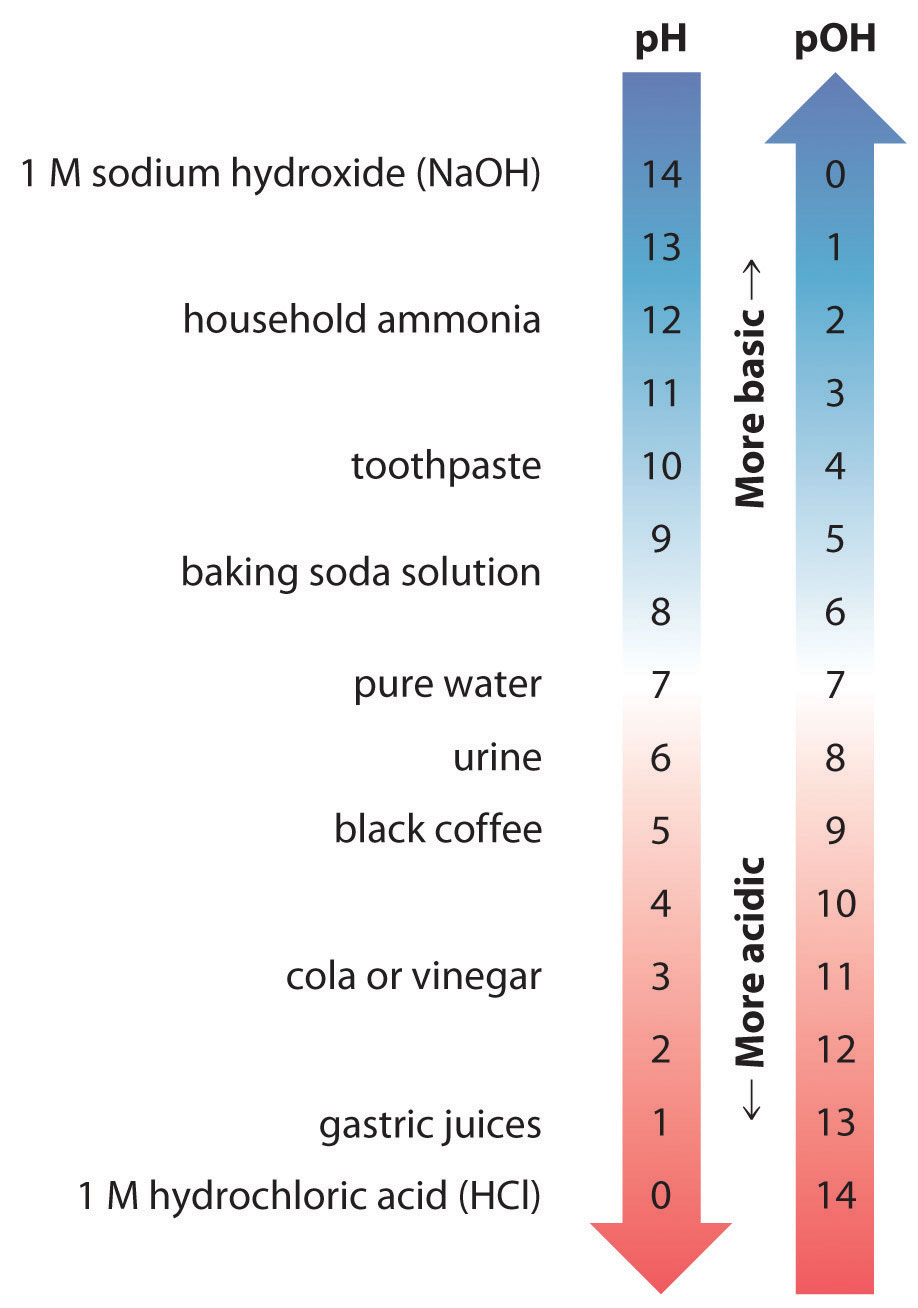
2.3 SelfIonization of Water and the pH Scale Chemistry LibreTexts

pH and pOH chart Diagram Quizlet
[Solved] . 8. Complete the chart PH POH Acid or Base H* Concentration
8.3 pH and pOH Chemistry for Chemical Engineers
Ph Definition Chemistry
PPT Chapter 17 Acids and Bases PowerPoint Presentation, free

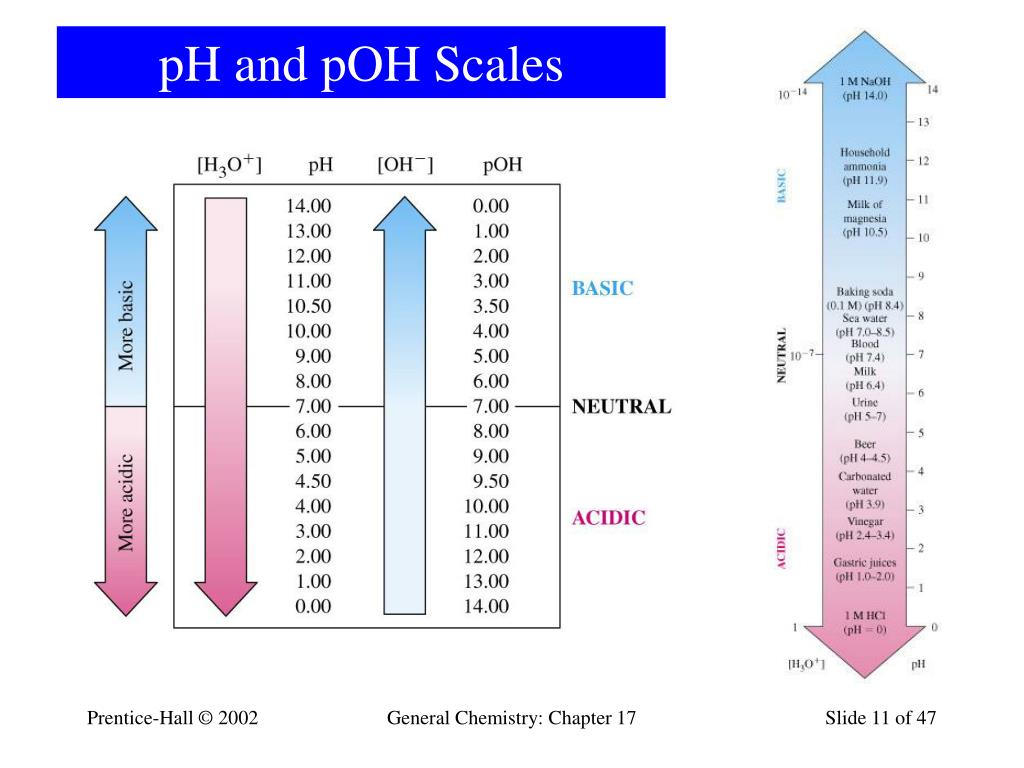
Ph And Poh Scale
PH pOH Chart PDF

Ph Poh Chart For Calculations
What is the pH Scale?, Examples, Video Chemistry Online
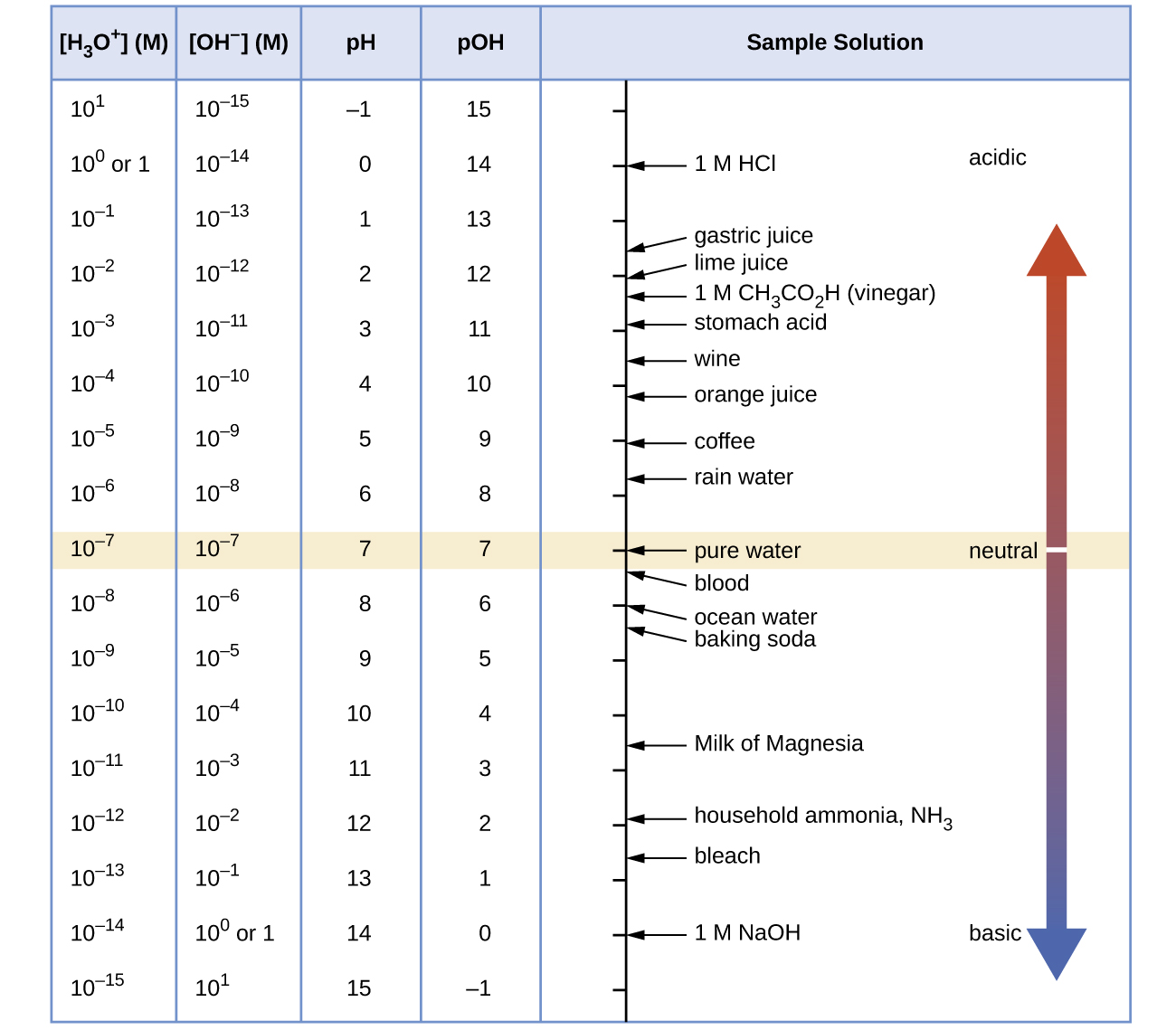
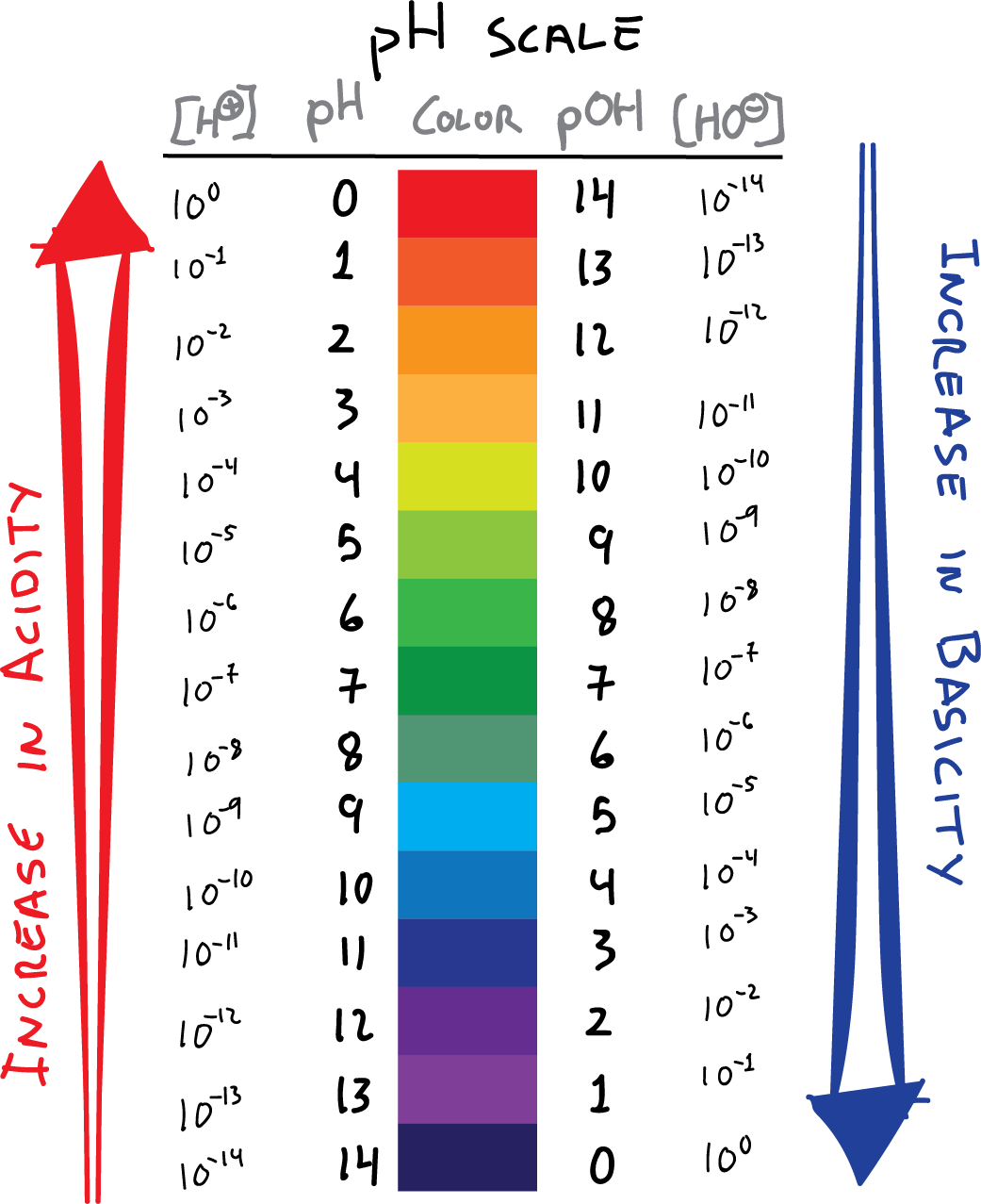
A Table Is Provided With 5 Columns.
Explain The Characterization Of Aqueous Solutions As Acidic, Basic, Or Neutral.
The First Column Is Labeled “Left Bracket H Subscript 3 O Superscript Plus Right Bracket (M).”
Web The Ph And Poh Values Of Some Common Substances At Standard Temperature (25 °C) Are Shown In This Chart.
Related Post:
