Salt Water Freezing Point Chart
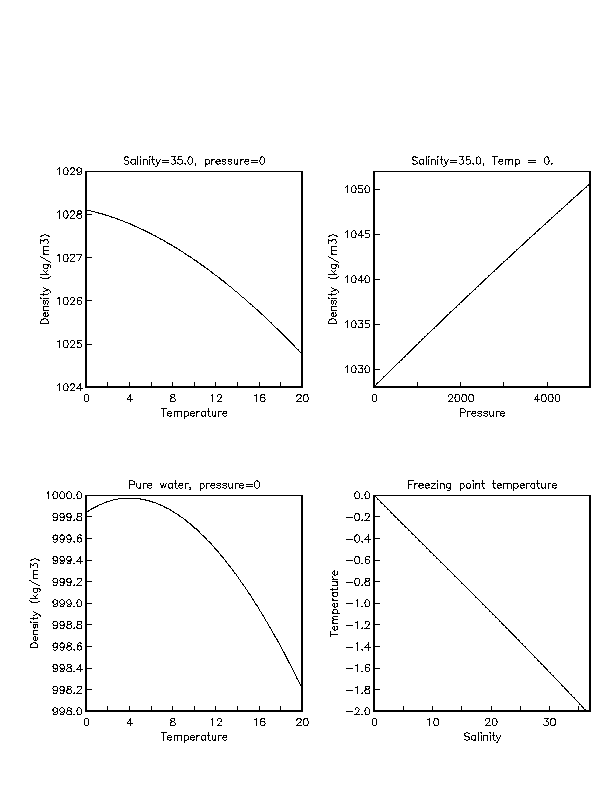
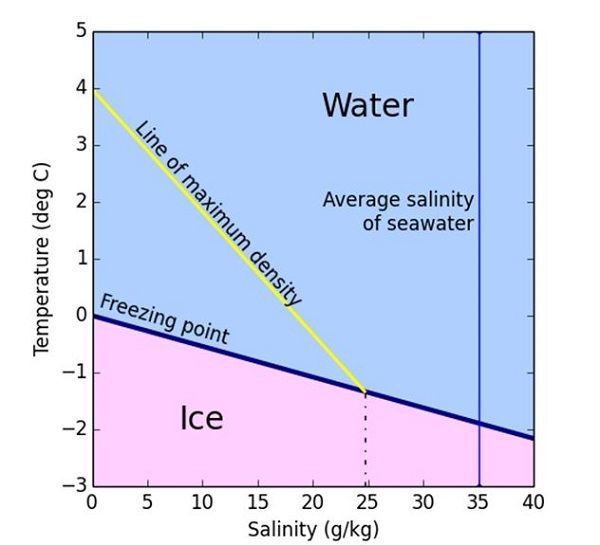
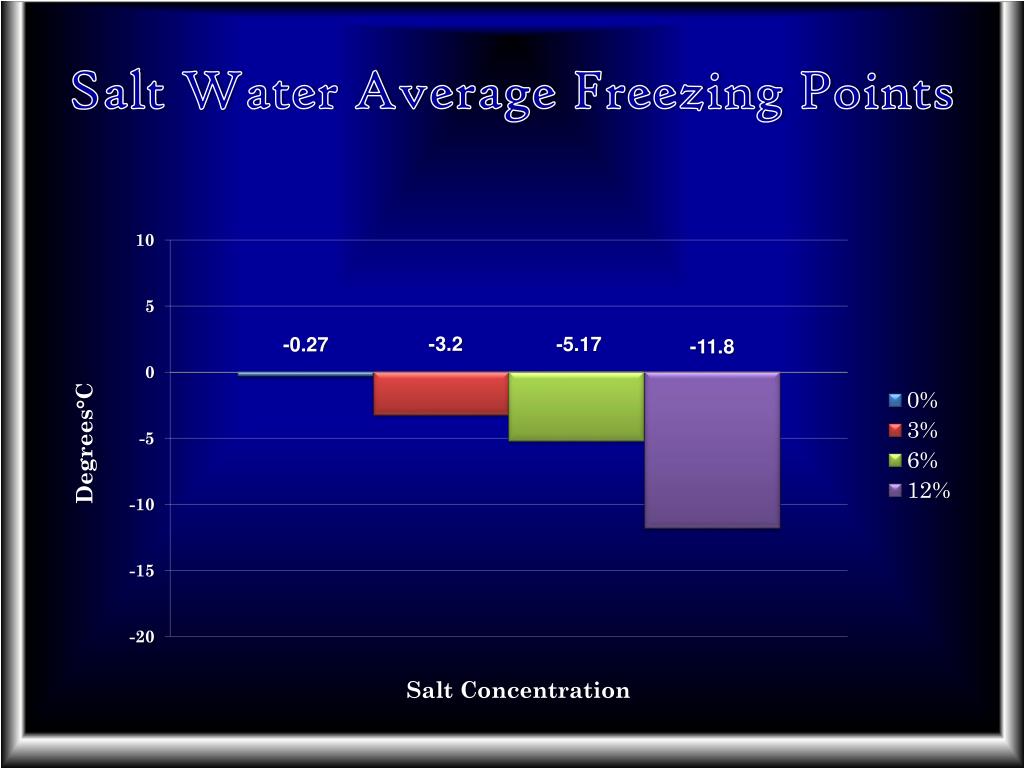
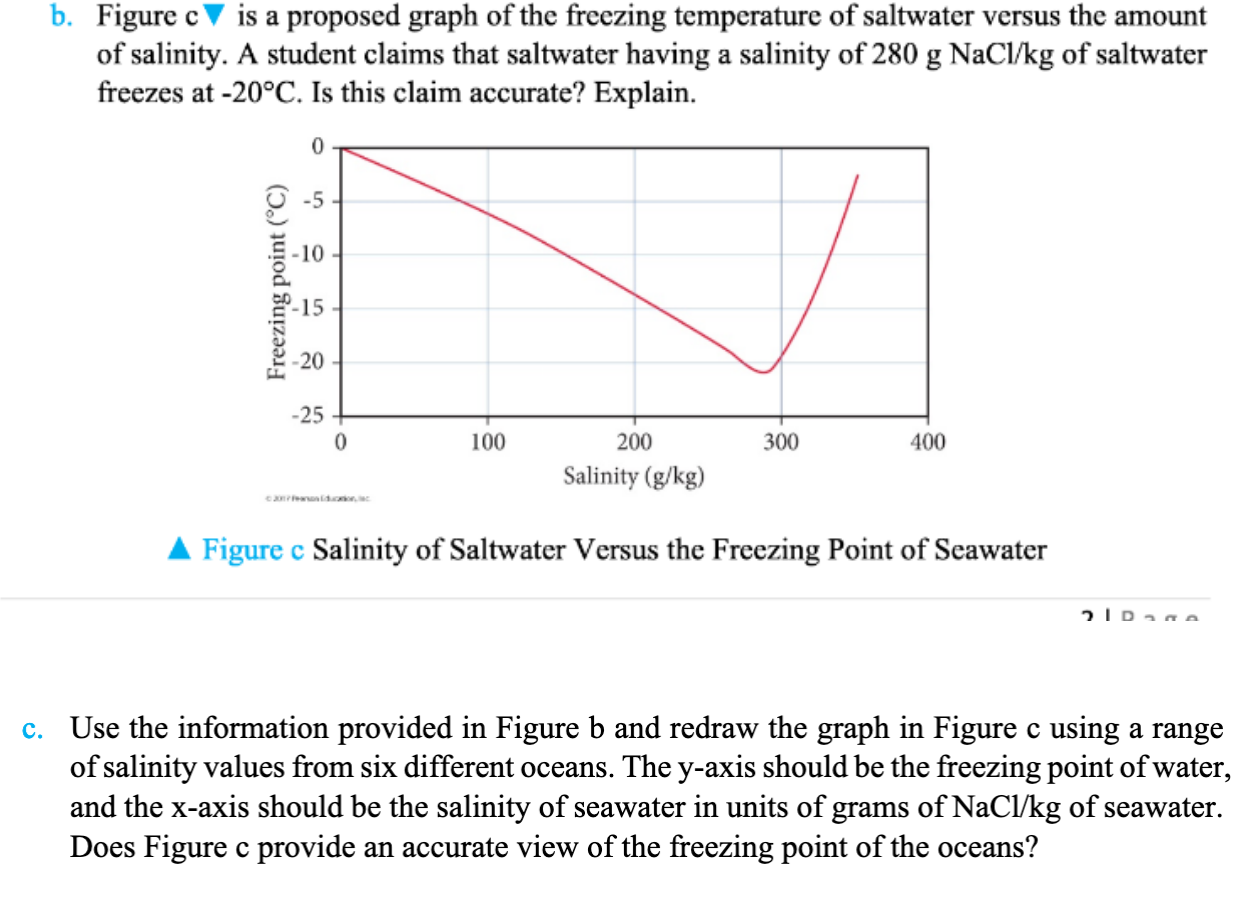
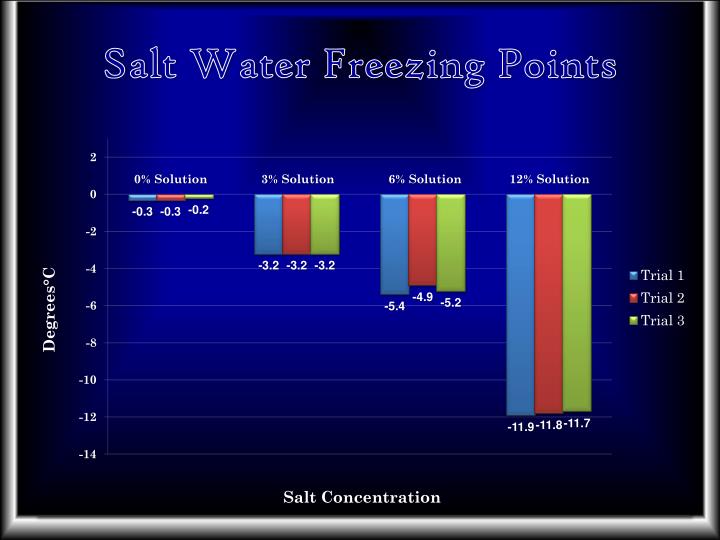
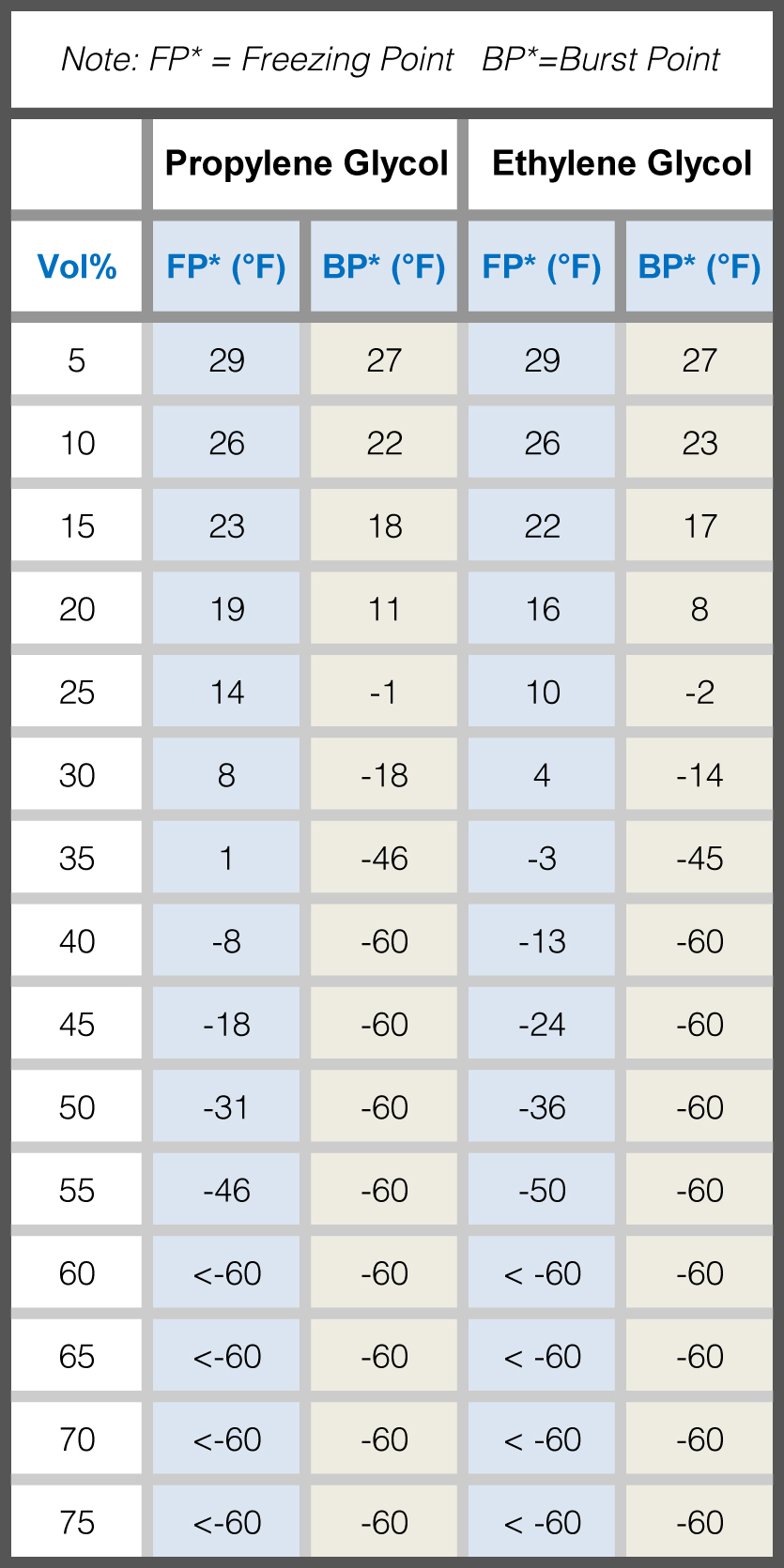
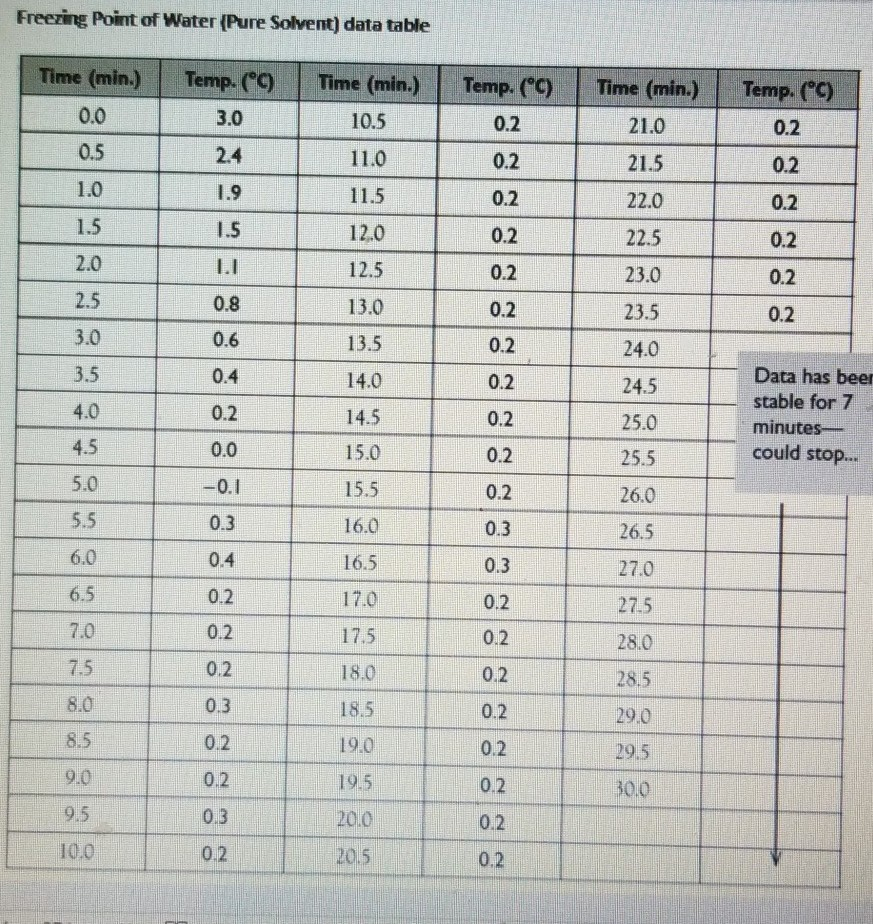
Salt Water Freezing Point Chart - You get the same effect if you add salt to ice cubes because the outer surface of ice always contains a thin film of water. Basically, the salt concentration in water (salinity) is. Web for saltwater that’s as saturated as it can possibly get (i.e. (initial observation) in winter time we spread salt in the roads and side walks to melt snow and ice or to prevent ice. But, solubility in a solvent also matters. Freezing point, density, specific heat and dynamic viscosity of sodium chloride and water coolant. Leveraging a precise formula, it offers insights into the properties and behaviors of saltwater under certain conditions. Web the freezing point of water is 32 degrees fahrenheit, 0 degrees celsius, and 273.15 kelvin. See also spfs calculator online. Do other material do the same? Popular internal searches in the engineering toolbox. How does salt melt ice or snow? Popular internal searches in the engineering toolbox. Web in water in which the concentration of salt is high, the freezing point can be as low as −6°c. This means that adding ice to winter roads can help water melt. Web the freezing point is the temperature at which crystals start to appear when you cool a liquid mixture. Web salt and the freezing point of water. When seawater freezes, however, the ice contains very little salt because only the water part freezes. S = 35000 mg/l = 35 ‰. Knowing how different chemicals affect water’s freezing point can help. The engineering toolbox privacy policy. Both are proportional to the molality of the solute. Web for saltwater that’s as saturated as it can possibly get (i.e. Do you know the freezing point of water? How does this melt ice? Web salt and the freezing point of water. Web the freezing point of a solution is always less than the freezing point of the pure solvent due to disruption of intermolecular interactions. Basically, the salt concentration in water (salinity) is. Leveraging a precise formula, it offers insights into the properties and behaviors of saltwater under certain conditions. Here is a. Web for saltwater that’s as saturated as it can possibly get (i.e. Do other material do the same? Proper brine concentration is important to fully submerge and freeze foods without issues. But, solubility in a solvent also matters. Web the document provides a table with the freezing point temperatures and corresponding salt concentrations in brine at 60°f. Web updated on may 05, 2019. But, solubility in a solvent also matters. Web salt and the freezing point of water. The engineering toolbox privacy policy. Is the freezing point the same as the melting point? You get the same effect if you add salt to ice cubes because the outer surface of ice always contains a thin film of water. Freezing point of seawater vs. How does salt melt ice or snow? This means that adding ice to winter roads can help water melt. Or are you a sailor who needs to determine if the. Electrical conductivity of seawater vs. At the eutectic point, the melting or freezing temperature is as low as it will get: How does this melt ice? Web salinity is normally quoted in units ‰ (parts per thousand): When seawater freezes, however, the ice contains very little salt because only the water part freezes. In the case of a salt solution with concentrations of salt greater than 23.3%, the solubility curve shows the temperature at which crystals of salt will appear when you cool a solution of a given concentration. Do other material do the same? Popular internal searches in the engineering toolbox. The engineering toolbox privacy policy. Web the freezing point of a. Web updated on may 05, 2019. The engineering toolbox privacy policy. Popular internal searches in the engineering toolbox. Or are you a sailor who needs to determine if the salt water in your boat's tanks is. Is salt creating heat to. The good news is you don't need a. Salt melts ice essentially because adding salt lowers the freezing point of the water. Freezing point, density, specific heat and dynamic viscosity of sodium chloride and water coolant. Web salinity is normally quoted in units ‰ (parts per thousand): How does salt melt ice or snow? Web the document provides a table with the freezing point temperatures and corresponding salt concentrations in brine at 60°f. Eventually, all the ice melts, leaving very cold salt water. At the eutectic point, the melting or freezing temperature is as low as it will get: Is salt creating heat to. But, solubility in a solvent also matters. Web salt and the freezing point of water. You get the same effect if you add salt to ice cubes because the outer surface of ice always contains a thin film of water. Are you planning to go on a beach vacation and wondering if you need to pack a wetsuit for the colder waters? The freezing point of saltwater is thus lower than the freezing point of pure water. Well, it doesn't, unless there is a little water available with the ice. How does this melt ice?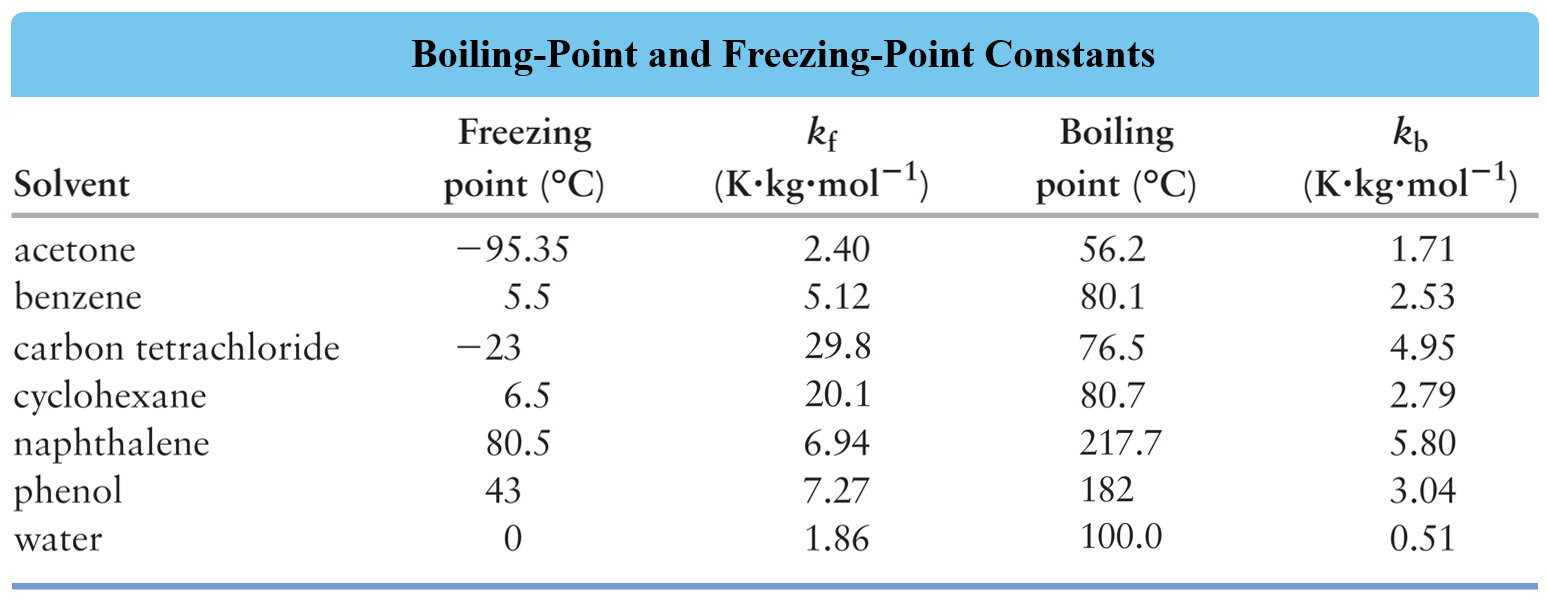
Freezing Point Depression Chemistry Steps

Understand how salt affects the freezing and melting points of water
freezing point of salt water karak
Sea ice an overview Met Office
PPT Salt Changes the Freezing Point of Water PowerPoint Presentation
37+ salt water freezing point calculator TameelSeraphina
PPT Salt Changes the Freezing Point of Water PowerPoint Presentation
Antifreeze Freezing Point Chart Online Shopping
Solved Freezing Point of Water (Pure Solvent) data table
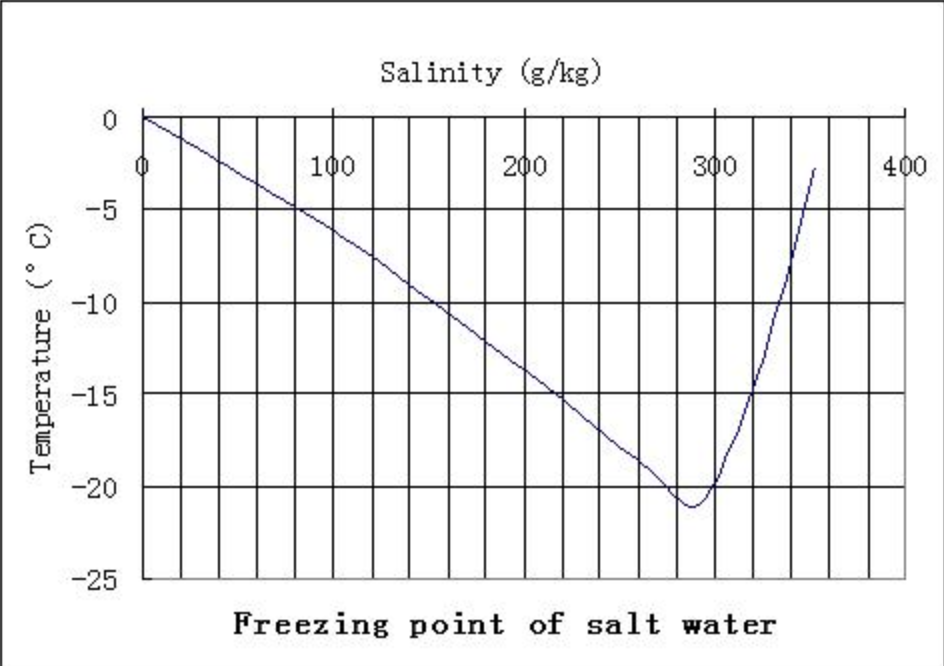
Physics 111 Fundamental Physics I The Physics of Salting Roads
When Seawater Freezes, However, The Ice Contains Very Little Salt Because Only The Water Part Freezes.
This Is When The Saltwater Is 23.3% Salt (By Weight).
Web The Boiling Point Is Raised By 0.5 Degrees Celsius For Water With 29.2 Grams Of Salt Dissolved In Each Kg Of Water.
The Engineering Toolbox Privacy Policy.
Related Post: