Vapor Pressure Of Water Chart
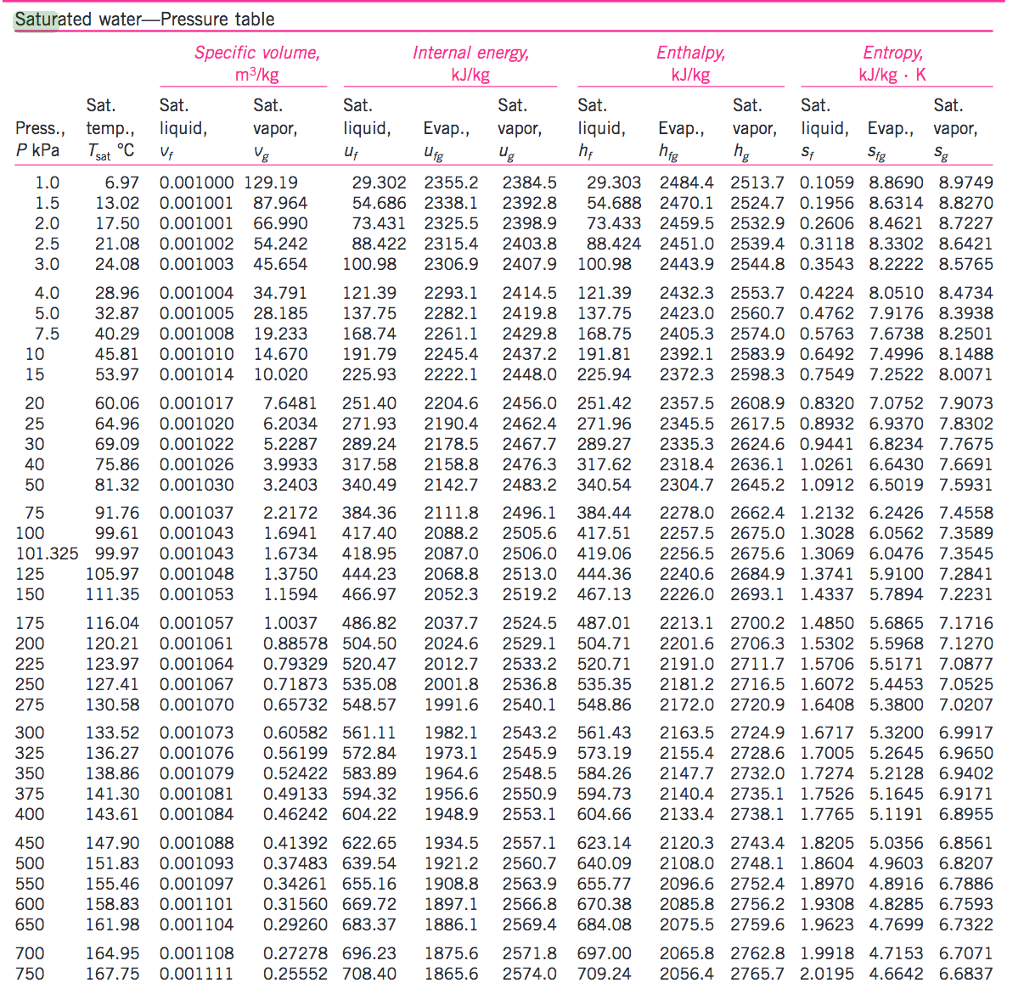
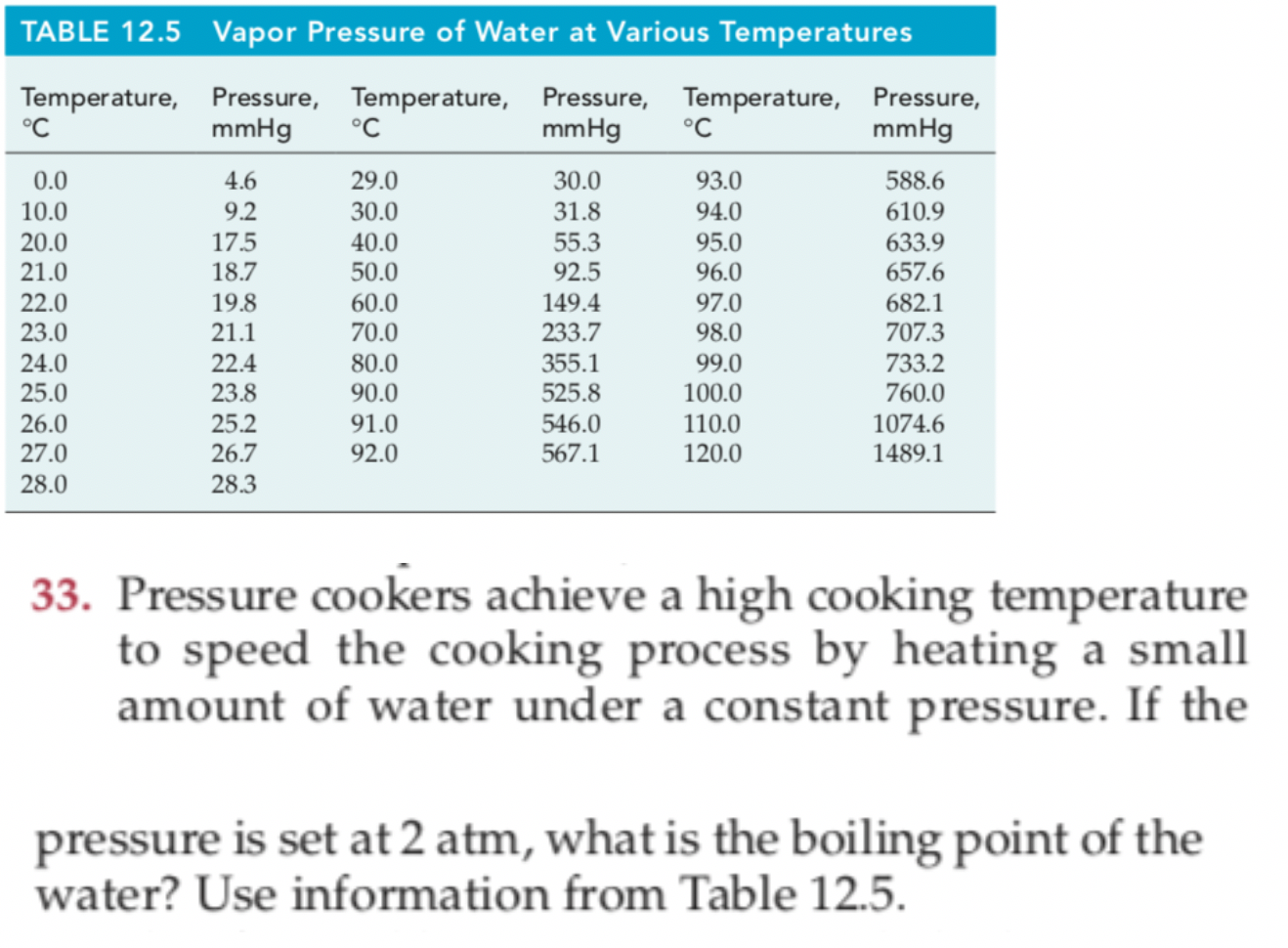
Vapor Pressure Of Water Chart - This chart shows that this trend is true for various substances with differing chemical properties. Web the vapor pressure of water is the pressure exerted by molecules of water vapor in gaseous form (whether pure or in a mixture with other gases such as air). For water, the vapor pressure reaches the standard sea level atmospheric pressure of 760 mmhg at 100°c. Web with this vapor pressure calculator, we present to you two vapor pressure equations! Web the boiling point is defined as the temperature at which the saturated vapor pressure of a liquid is equal to the surrounding atmospheric pressure. And what does this all have to do with. Using this 1st calculator, you insert temperature in °f, and get the vapor pressure of water in terms of kpa, psi, mmhg, bar, atm, torr. Web the vapor pressure of water at room temperature (25 ° c) is 23.8 mm hg, 0.0313 atm, or 23.8 torr, or 3.17 kpa. Vapor pressure is directly proportional to temperature). You can see the drinking duck in action in the video below: Web the boiling point is defined as the temperature at which the saturated vapor pressure of a liquid is equal to the surrounding atmospheric pressure. 1 atmosphere pressure is 101.325 kpa. It is the pressure exerted by the saturated vapour in contact with the surface of the liquid at that temperature. Have you found yourself wondering: ” crc handbook of. Web to understand that the equilibrium vapor pressure of a liquid depends on the temperature and the intermolecular forces present. Web vapor pressure [a] or equilibrium vapor pressure is the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases (solid or liquid) at a given temperature in a closed system. For water, the vapor pressure reaches the. For water, the vapor pressure reaches the standard sea level atmospheric pressure of 760 mmhg at 100°c. Copyright © 2024 claude yoder. Web the vapor pressure of water calculator is a handy tool that can help in determining the vapor pressure of water and ice. ” crc handbook of chemistry and physics,. Vapor pressure of water is shared under a. Web with this vapor pressure calculator, we present to you two vapor pressure equations! Web the vapor pressure of water calculator is a handy tool that can help in determining the vapor pressure of water and ice. Web water vapor pressure chart. Web the vapor pressure of water at room temperature (25 ° c) is 23.8 mm hg, 0.0313 atm,. The equilibrium vapor pressure is an indication of a liquid's thermodynamic tendency to evaporate. Web to understand that the equilibrium vapor pressure of a liquid depends on the temperature and the intermolecular forces present. The pressure exerted by the vapor phase is called the. Note that when water vapor pressure equals atmospheric pressure, then the water molecules are free to. Note that when water vapor pressure equals atmospheric pressure, then the water molecules are free to jump into the gas state. If you're unsure what vapor pressure is, keep scrolling. At its freezing point (0 ° c), the vapor pressure of water is 4.6 torr. Using this 1st calculator, you insert temperature in °f, and get the vapor pressure of. Web below are some selected values of temperature and the saturated vapor pressures required to place the boiling point at those temperatures. That is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample of the liquid (or solid) in a closed container. The equilibrium vapor pressure is an indication of a liquid's thermodynamic. Nist chemistry webbook, srd 69. The saturation vapor pressure is the pressure at which water vapor is in thermodynamic equilibrium with its condensed state. The pressure exerted by the vapor phase is called the. For water, the vapor pressure reaches the standard sea level atmospheric pressure of 760 mmhg at 100°c. Web the vapor pressure of water at room temperature. The pressure up cancels the pressure down and boiling begins. Using this 1st calculator, you insert temperature in °f, and get the vapor pressure of water in terms of kpa, psi, mmhg, bar, atm, torr. Web the vapor pressure of water calculator is a handy tool that can help in determining the vapor pressure of water and ice. It is. Using this 1st calculator, you insert temperature in °f, and get the vapor pressure of water in terms of kpa, psi, mmhg, bar, atm, torr. Web the vapor pressure of a liquid is defined as the pressure exerted by the molecules that escapes from the liquid to form a separate vapor phase above the liquid surface. Water vapour pressure table. Vapor pressure of h 2 o at various temperatures (celsius) modified from nebergall et. Web to understand that the equilibrium vapor pressure of a liquid depends on the temperature and the intermolecular forces present. Vapor or saturation pressure depends on temperature. Web the graph of the vapor pressure of water versus temperature in figure \(\pageindex{3}\) indicates that the vapor pressure of water is 68 kpa at about 90 °c. To understand that the relationship between pressure, enthalpy of vaporization, and temperature is given. Note that when water vapor pressure equals atmospheric pressure, then the water molecules are free to jump into the gas state. The motion of the duck illustrates a physical principle called vapor pressure. If you're unsure what vapor pressure is, keep scrolling. Web the vapor pressure of a liquid is defined as the pressure exerted by the molecules that escapes from the liquid to form a separate vapor phase above the liquid surface. Vapor pressure of water is shared under a not declared license and was authored, remixed, and/or curated by libretexts. At its boiling point (100 ° c), the vapor pressure of water is 658.0 torr (atmospheric pressure). Web from crc handbook of chemistry and physics, 65th edition (rounded to two decimal places) temp, °c. It is the pressure exerted by the saturated vapour in contact with the surface of the liquid at that temperature. Web the vapor pressure of water at room temperature (25 ° c) is 23.8 mm hg, 0.0313 atm, or 23.8 torr, or 3.17 kpa. ” crc handbook of chemistry and physics,. Using this 1st calculator, you insert temperature in °f, and get the vapor pressure of water in terms of kpa, psi, mmhg, bar, atm, torr.
Table Of Vapor Pressure Of Water A Guide To Understanding Its

Conservation physics Fundamental microclimate concepts

Vapor Pressure Chart For Water

vapor pressure of water table
Vapour Pressure Of Water Chart

Water Vapour Pressure Chart Bar
Vapor Pressure Chart For Water
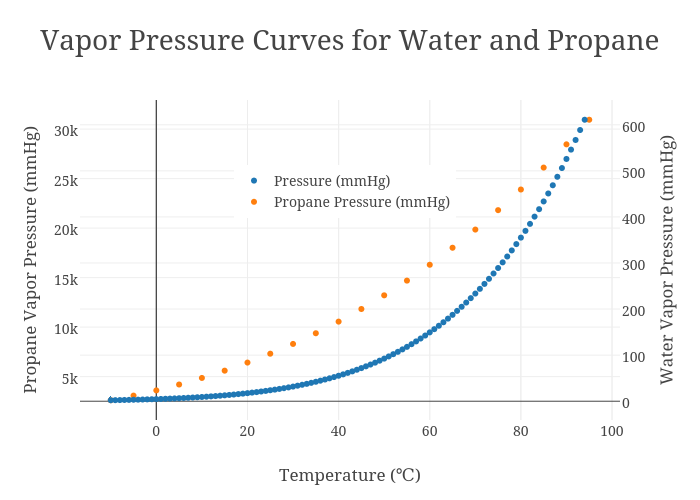
Vapor Pressure Curves for Water and Propane scatter chart made by

Vapor Pressure Chart For Water
Chapter 11.4 Vapor Pressure Chemistry LibreTexts
Web Vapor Pressure [A] Or Equilibrium Vapor Pressure Is The Pressure Exerted By A Vapor In Thermodynamic Equilibrium With Its Condensed Phases (Solid Or Liquid) At A Given Temperature In A Closed System.
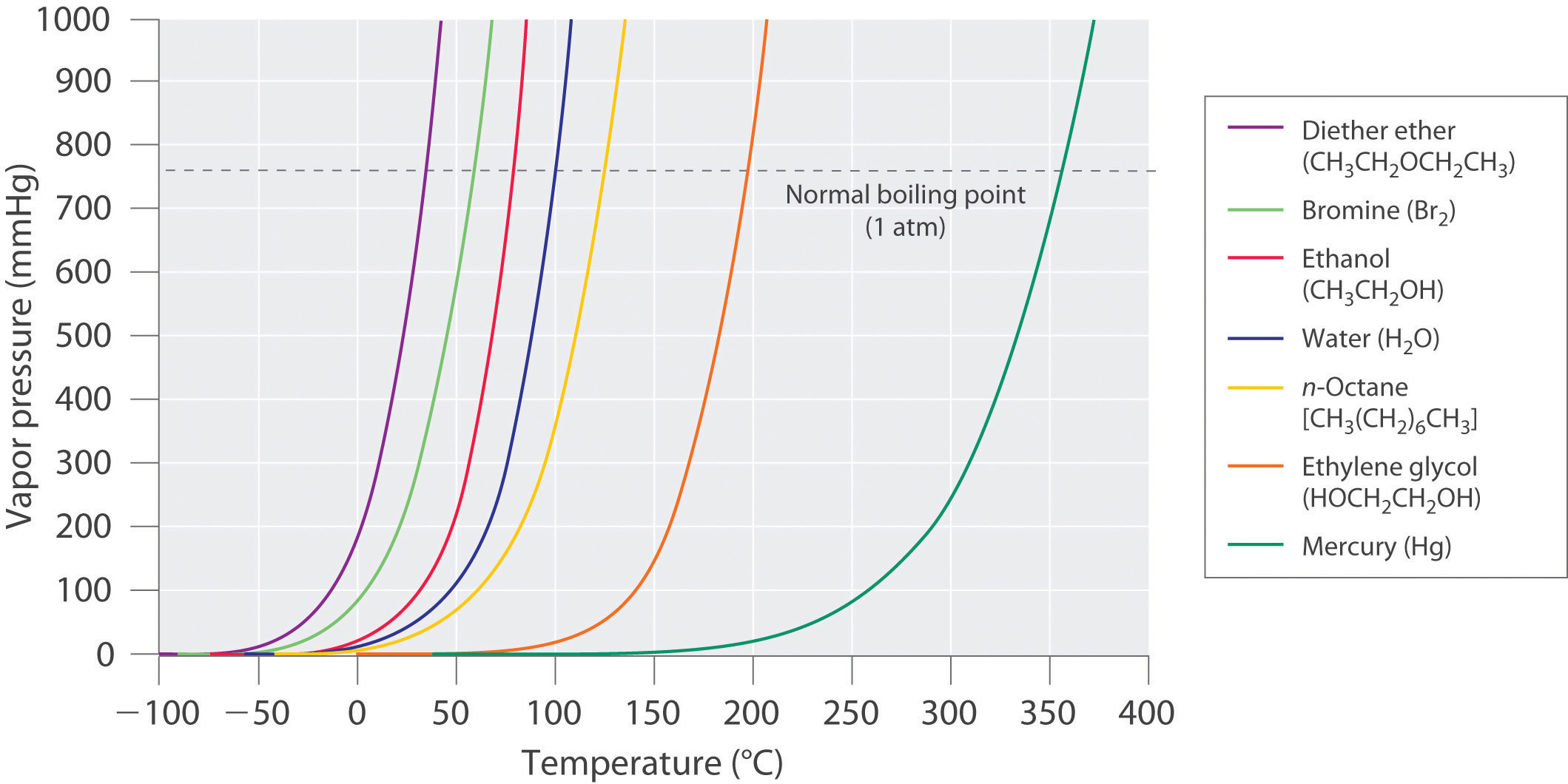
Web The Graph Shows How The Saturated Vapor Pressure (Svp) Of Water Varies From 0°C To 100 °C.
Web The Vapor Pressure Of A Liquid Is The Equilibrium Pressure Of A Vapor Above Its Liquid (Or Solid);
The Pressure Up Cancels The Pressure Down And Boiling Begins.
Related Post: